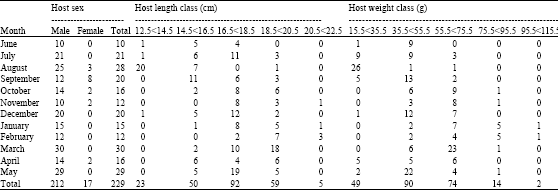
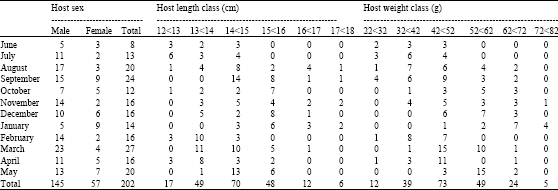
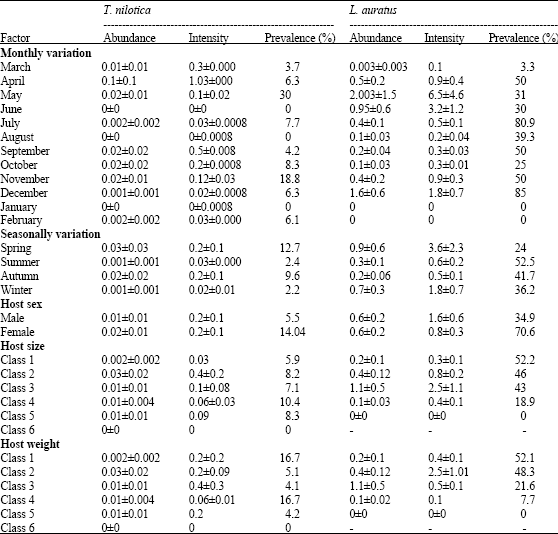
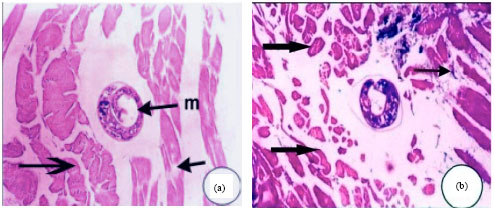
Research Article
Responses of the Mullet, Liza auratus and the Cichlid, Oreochromis niloticus from Lake Manzala (Egypt) to Heterophyd Infection
Department of Zoology, Faculty of Science, Suez Canal University, Ismailia, Egypt
M. F.M. Soliman
Department of Zoology, Faculty of Science, Suez Canal University, Ismailia, Egypt
E. A. Hassan
Department of Zoology, Faculty of Science, Suez Canal University, Ismailia, Egypt