Research Article
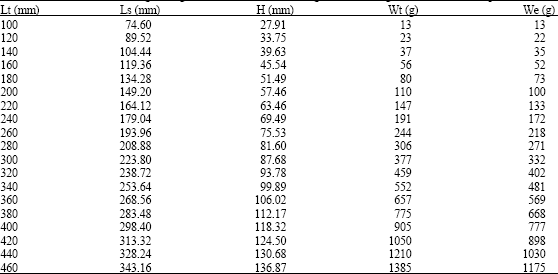
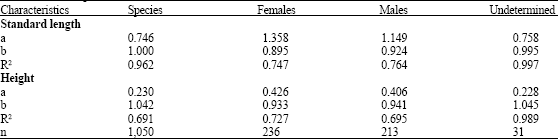
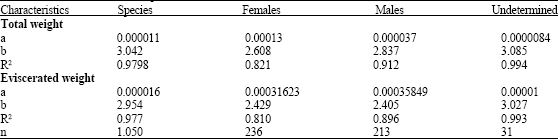
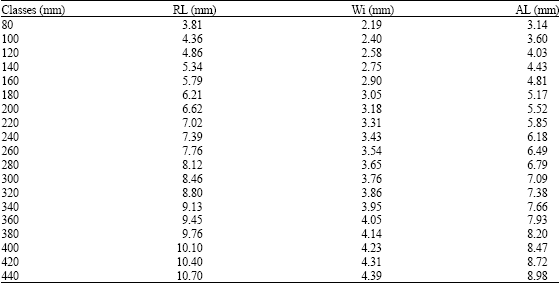
Age Determination of Microlepidotus brevipinnis (Steindachner, 1869) (Pisces: Haemulidae) in the Coast of Jalisco, Mexico, by Reading Otoliths and Scales
Instituto de Ciencias Del Mar y Limnologia, UNAM, Apartado Postal 70-305, Mexico D.F., 09340, Mexico
M. Gallardo- Cabello
Instituto de Ciencias Del Mar y Limnologia, UNAM, Apartado Postal 70-305, Mexico D.F., 09340, Mexico
E. Espino- Barr
CRIP-Manzanillo, INAPESCA, Playa Ventanas s/n, Manzanillo, Colima, 28200, Mexico
E.G. Cabral- Solis
CRIP-Manzanillo, INP, Playa Ventanas s/n, Manzanillo, Colima, 28200, Mexico