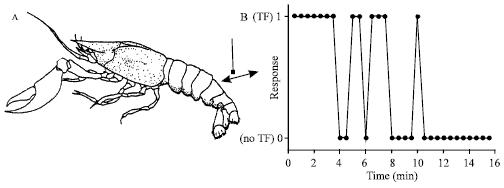
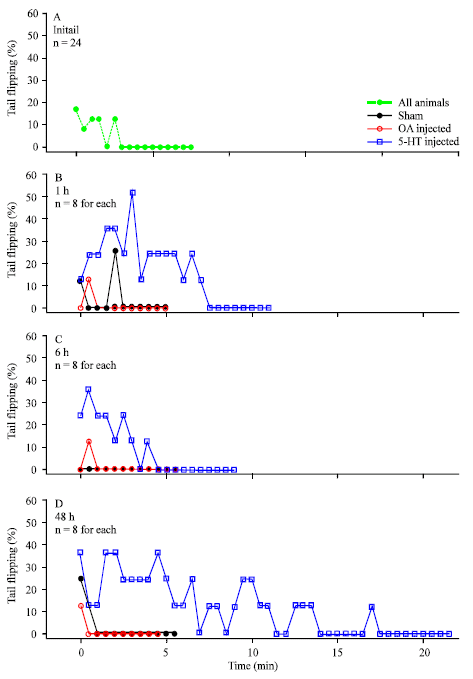
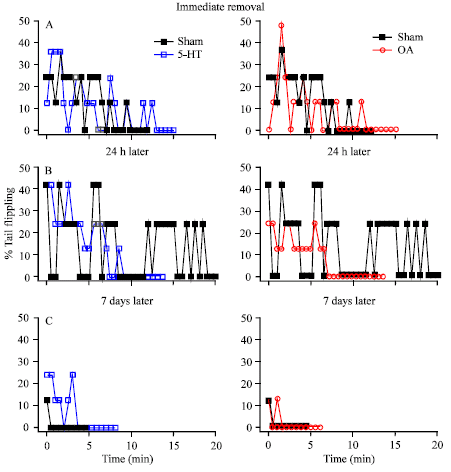
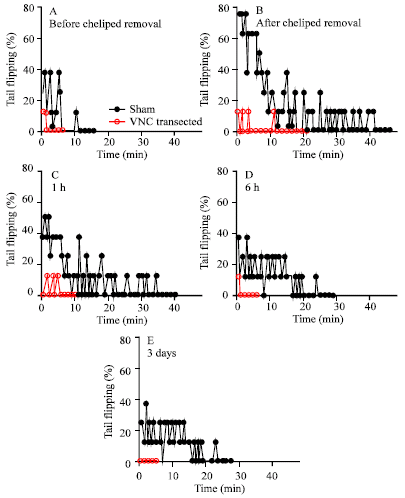
Research Article
Modification of the Tail Flip Escape Response in Crayfish by Neuromodulation and Behavioral State with and without Descending CNS Input
Department of Biology, University of Kentucky, Lexington, KY USA, 40506-0225
Walter Hailes
Department of Biology, University of Kentucky, Lexington, KY USA, 40506-0225
Robin L. Cooper
Department of Biology, University of Kentucky, Lexington, KY USA, 40506-0225