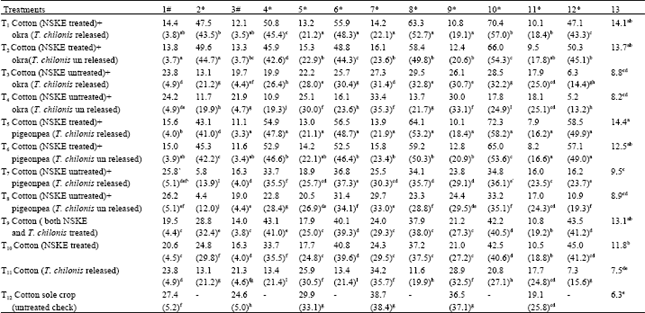
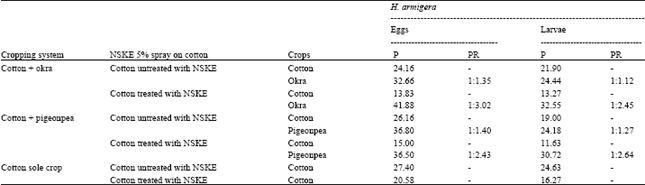
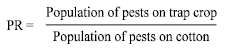Research Article
Mitigation of Insecticide Resistance in Helicoverpa armigera (Hubner) (Lepidoptera:Noctuidae) by Conjunctive Use of Trap Crops, Neem and Trichogramma chilonis Ishii in Cotton
Department of Agricultural Entomology, Tamil Nadu Agricultural University, Coimbatore-641 (003, India
A. Regupathy
Department of Agricultural Entomology, Tamil Nadu Agricultural University, Coimbatore-641 (003, India