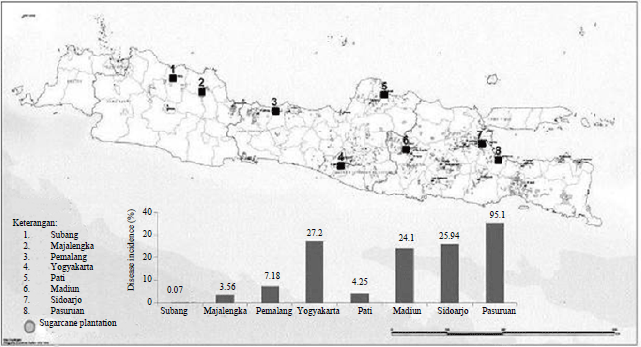
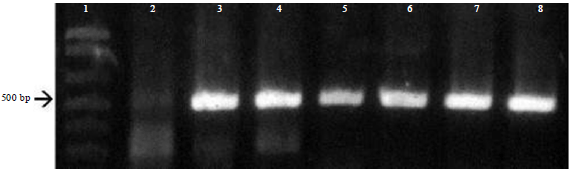
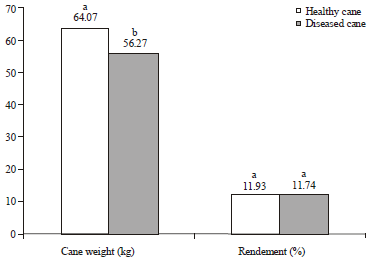
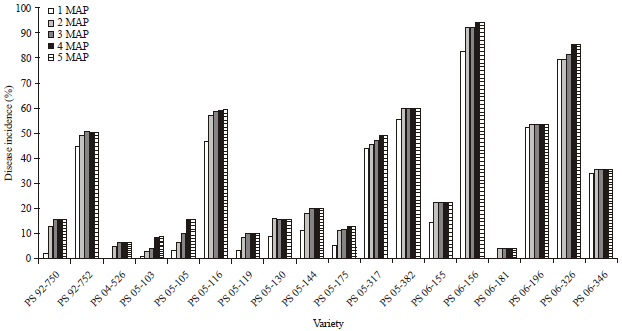
Research Article
Dispersal, Yield Losses and Varietal Resistance of Sugarcane streak mosaic virus (SCSMV) in Indonesia
Post Graduate Program, Faculty of Agriculture, Brawijaya University, Jl. Veteran Malang, 65145, Indonesia
Tutung Hadi Astono
Indonesia Sugar Research Institute, Jl. Pahlawan 25 Pasuruan, 67126, Indonesia
Siti Rasminah Ch. Syamsidi
Indonesia Sugar Research Institute, Jl. Pahlawan 25 Pasuruan, 67126, Indonesia
Syamsudin Djauhari
Indonesia Sugar Research Institute, Jl. Pahlawan 25 Pasuruan, 67126, Indonesia