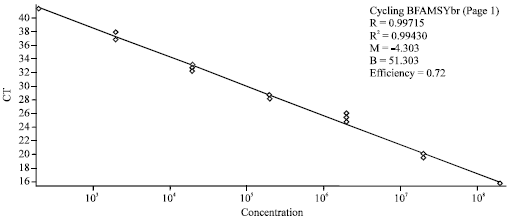
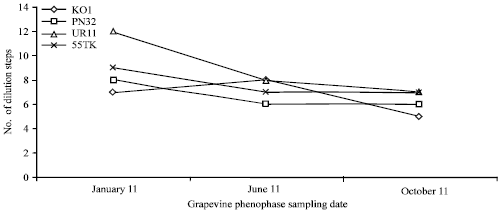
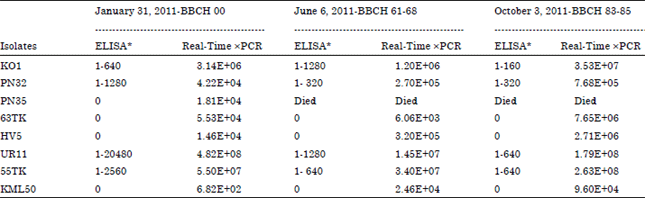
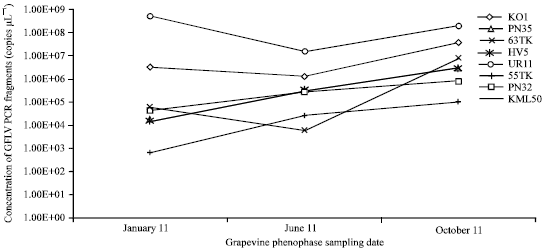
Research Article
Detection of Moravian Isolates of GFLV: Comparison of Real-Time RT-PCR and ELISA
Mendel University, Faculty of Horticulture, Mendeleum Institute of Genetics and Plant Breeding, Valticka 334, 691 44, Lednice, Czech Republic
Petr Kominek
Crop Research Institute, Drnovsk� 507, 161 06 Prague 6-Ruzyn, Czech Republic