Research Article
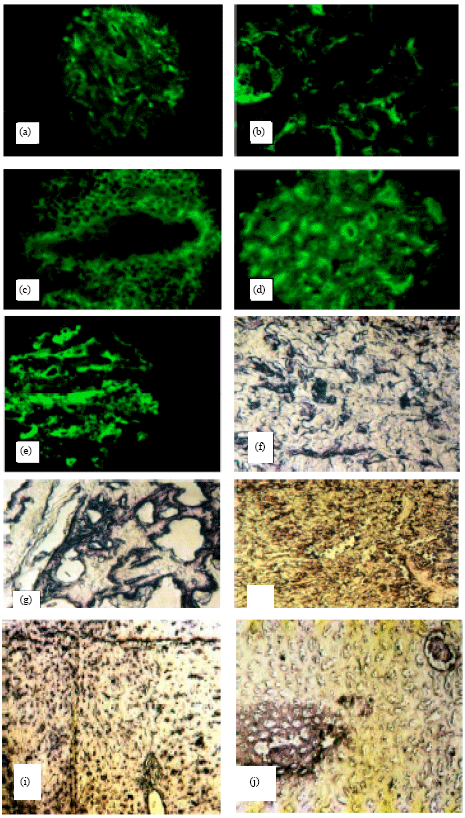
Lumpy Skin Disease Virus Identification in Different Tissues of Naturally Infected Cattle and Chorioallantoic Membrane of Emberyonated Chicken Eggs Using Immunofluorescence, Immunoperoxidase Techniques and Polymerase Chain Reaction
Department of Virology, Faculty of Veterinary Medicine, Mansoura University, Egypt
M.S. El-Tholoth
Department of Virology, Faculty of Veterinary Medicine, Mansoura University, Egypt