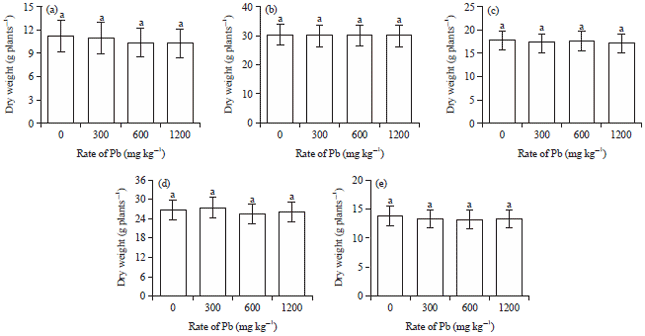
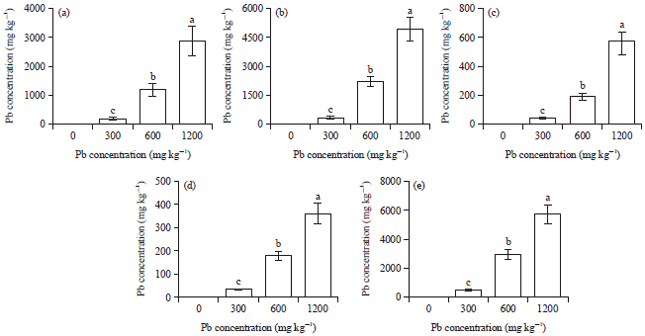
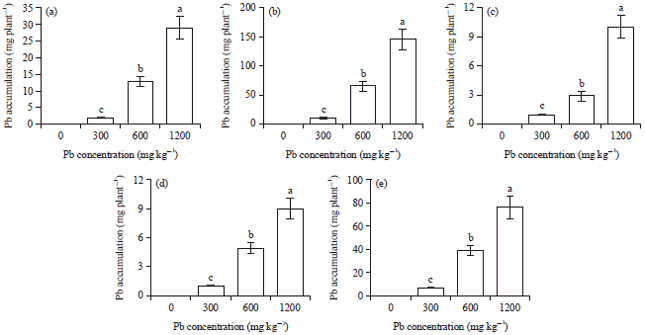
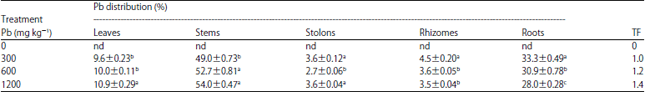
Research Article
Phytoextraction Efficiency of Lead by Arum (Colocasia esculenta L.) Grown in Soil
Department of Soil Science, University of Chittagong, 4331 Chittagong, Bangladesh
LiveDNA: 880.15653
Md. Abul Kashem
Department of Soil Science, University of Chittagong, 4331 Chittagong, Bangladesh
Khan Towhid Osman
Department of Soil Science, University of Chittagong, 4331 Chittagong, Bangladesh