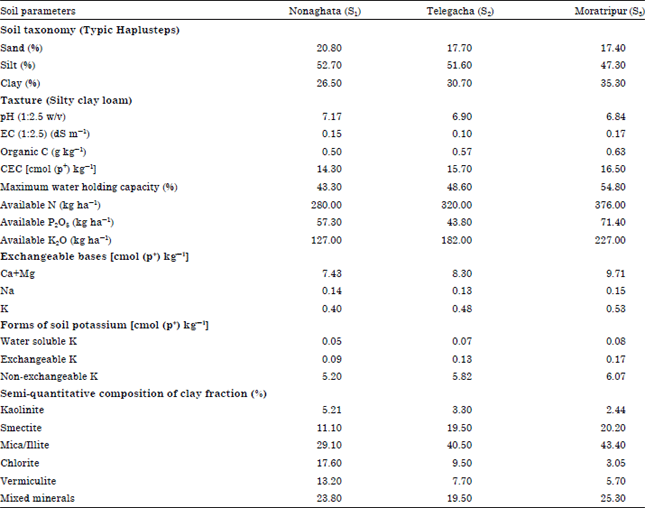
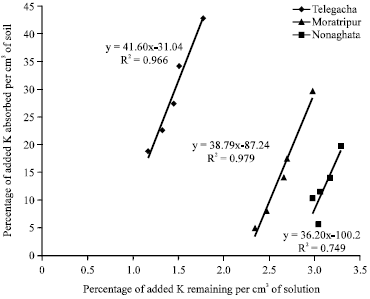
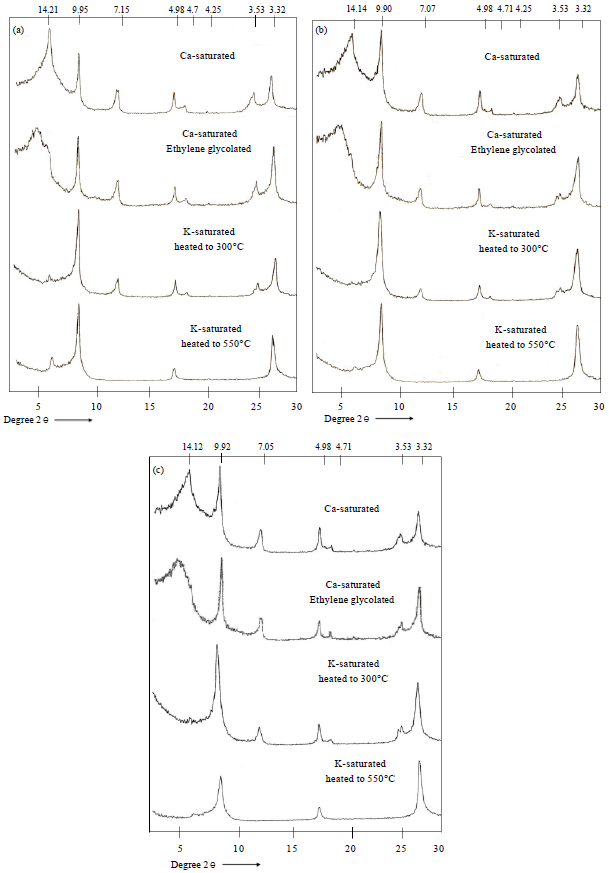
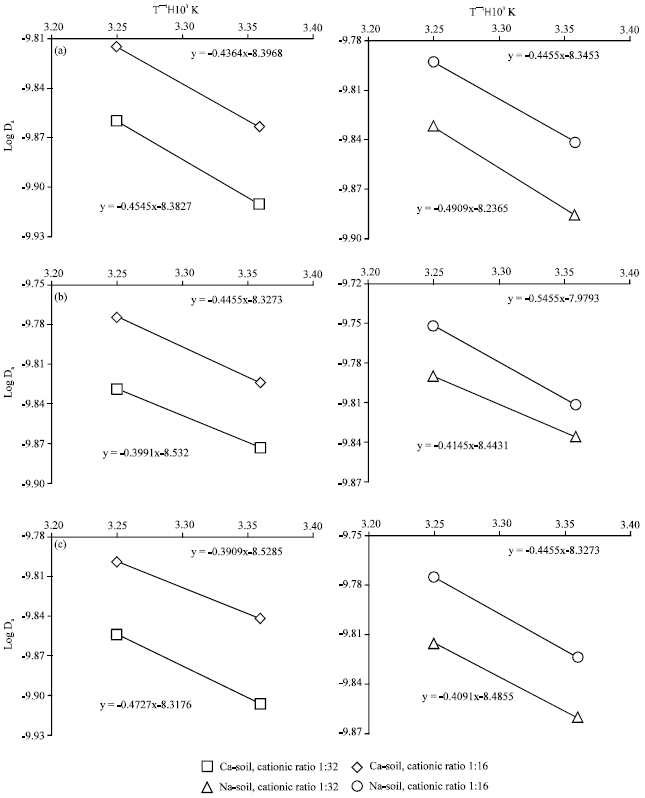
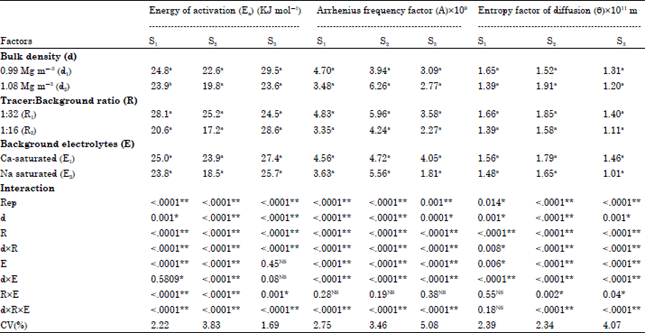
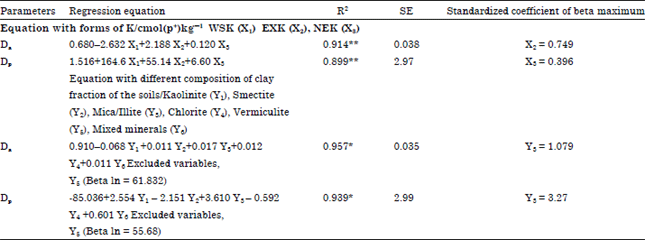
Research Article
Effect of Different Factors on Diffusion Characteristics of Potassium in Alluvial Soils of Eastern Indo Gangetic Plain
Department of Agricultural Chemistry and Soil Science, Bidhan Chandra Krishi Viswavidyalaya, Mohanpur, 741252, Nadia, West Bengal, India
Debjani Ghosh
Department of Agricultural Chemistry and Soil Science, Bidhan Chandra Krishi Viswavidyalaya, Mohanpur, 741252, Nadia, West Bengal, India