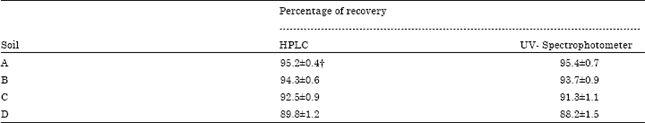
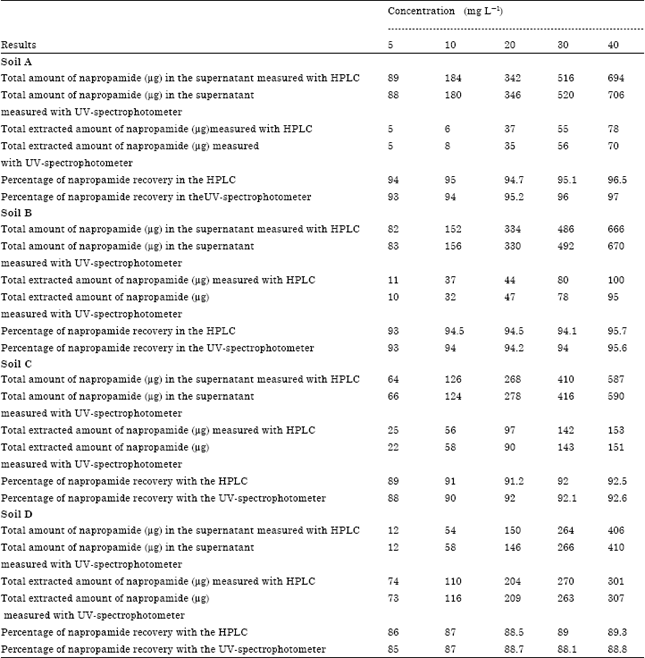
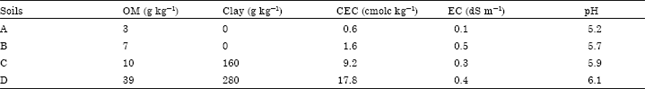
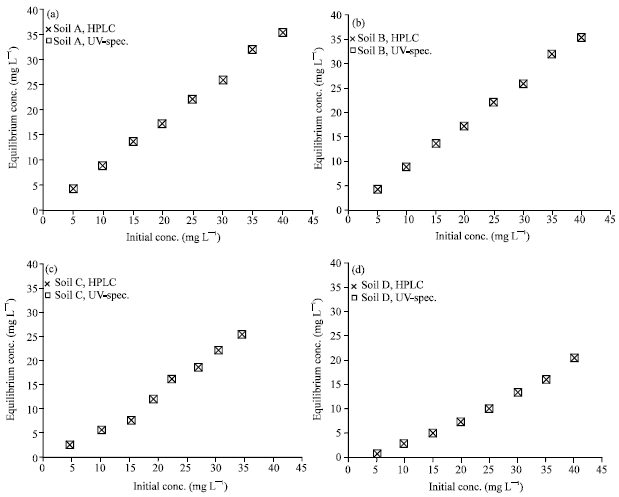
Research Article
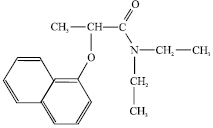
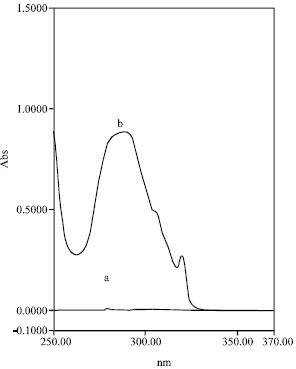
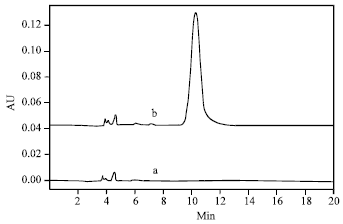
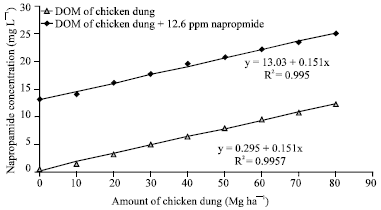
Comparison between UV-spectrophotometry and HPLC Methods to Determine Napropamide Concentration in Soil Sorption Experiment
Department of Land Management, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
Samsuri Abd Wahid
Department of Land Management, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
Dzolkhifli Omar
Department of Plant Protection, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
Radziah Othman
Department of Land Management, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
Bahi Jalili Seh-Bardan
Department of Land Management, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
Nur- Aisyah Sajudi
Department of Plant Protection, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia