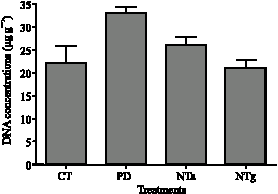
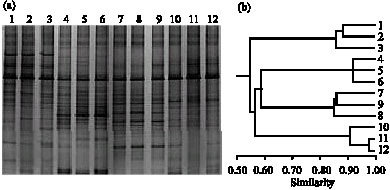
Research Article
Assessing Microbial Community in Andisol Differing in Management Practices by Biochemical and Molecular Fingerprinting Methods
Department of Soil Science, National University, Gazipur, Bangladesh
Atsushi Okubo
Faculty of Agriculture and Life Science, Hirosaki University, Hirosaki 036-8156, Japan
Shigenao Kawai
Faculty of Agriculture, Iwate University, Morioka 020-8550, Japan
Shu-ichi Sugiyama
Faculty of Agriculture and Life Science, Hirosaki University, Hirosaki 036-8156, Japan