ABSTRACT
This study investigated the use of Isotope Exchange Kinetics (IEK) technique to estimate lability and temporal variability of cadmium (Cd) in arable soils proximal to an automobile service centre at Owerri, south-eastern Nigeria. Thirty topsoil samples (0-10 cm depth) were collected from 5 arable farms receiving waste materials from automobile servicing. The samples were subjected to experimental conditions to determine exchangeable Cd using the IEK technique. There were slight to moderate variation in the distribution of soil exchangeable Cd among farms. Results also showed that isotopically exchangeable Cd (E(t)) predicted exchangeability of Cd up to 24 h of exchange from a short-term isotopic kinetics, that is, at >60 min. There was significant positive relationship r(I)/R (ratio of the radioactivity remaining in solution after 1 min of exchangeable to the total introduced radioactivity R) and exchangeable Cd (r = 0.8**; p<0.01). Interestingly, there was no significant relationship between soil pH and kinetic parameters.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijss.2007.48.54
URL: https://scialert.net/abstract/?doi=ijss.2007.48.54
INTRODUCTION
Heavy metals appear naturally in soils (Ojanuga et al., 1996) but at certain levels of bioaccumulation, they become toxic to living components of the ecosystem. With the exception of vanadium and titanium used by animals in their metabolic process (Manahan, 1994), most heavy metals are associated with profound health hazards. However, their impact depends on source as metal forms from anthropogenic sources are environmentally unstable, more soluble and bio-available than natural sources (Naidu et al., 1997).
Heavy metals occur in a variety of physicochemical forms, namely free or complexed ions in soil solution, adsorbed at the surfaces of clays, Fe and Mn oxyhdroxides or organic matter that are easily exchangeable, present in the lattice secondary minerals (phosphates, sulphides, carbonates), occluded in amorphous materials and present in crystal lattices of primary mineral (Berti and Jacobs, 1996). In all these, the most labile and bioavailable are those extracted in soil solution and adsorbed (Ciba et al., 1999).
Cadmium (Cd) is a bioavailable and biotoxic heavy metal regarded as an important environmental pollutant in agricultural soils. It is the labile fraction rather the total soil Cd which is critical when assessing Cd availability in soils (Gray et al., 2004).
The Isotope Exchange Kinetics (IEK) technique has been used to estimate rate and quantity of transfer of 32PO4 ions from soil solution to the solid phase (Frossard and Sinaj, 1997). The IEK was also applied in the study of the exchangeability of zinc in soils (Sinaj et al., 1999) as well as the assessment of phyto-availability of nickel in nickel-polluted soils.
The major objective of this study was to assess cadmium distribution in arable soils using Isotope Exchange Kinetics as well as predict temporal variability in its exchangeability at short-term (<60 min) and long-term (18 days) exchange intervals.
MATERIALS AND METHODS
Soil Sampling
Thirty topsoil samples (0-20 cm) were collected from 6 arable–farm locations receiving automobile wastes at an Automobile Service Centre in Owerri South-eastern Nigeria. On each farm 5 topsoil samples were collected. The soil samples were air-dried and sieved to obtain the <2 mm fraction before laboratory analysis.
Laboratory Analysis
Soil texture was determined by hydrometer method (Gee and Or, 2002). Soil pH was estimated electrometrically on a 1:2 soil/water solution (Hendershort et al., 1993). Total carbon was measured by LECO CNS 2000 analyzer (LECO, Australia). Cation exchange capacity (CEC) was determined by ammonium acetate leaching at pH 7.0 (Blakemore et al., 1987).
Total soil cadmium was determined by digestion in 5 mL H2O2 and 5 mL HNO3 for 5 h according to the procedure of Kovacs et al. (2000) and made to mark with 50 mL de-ionized water before filtered through Whatman 42 filter paper. Total Cd was measured in the digest by graphite furnace atomic absorption spectrometry (GFAAS) with deuterium arc background correlation and 0.1% (v/V) H3PO4 acid used as a modifier. Ethylene diamine-tetra-acetic acid (EDTA) extractable Cd was determined by extracting 5 g of soil with 20 mL of 0.04 EDTA at 20°C for 2 h on an end-over-end shaker. After extraction the suspension was centrifuged at 9400x g for 10 min and filtered through Whatman 42 filter paper before analysis of Cd by Flame Atomic Absorption Specrophometry (FAAS) with deuterium arc background correction.
Cadmium availability in soils were estimated by the Isotope Exchange Kinetics (IEK) on 5 soil samples. In this technique, 10 g of soil was equilibrated with 99 mL of de-ionized water on an end-over-end shaker for 5 days. This represented the mixing interval used to equilibrate the soil solution. After this time, at t = 0, 1 mL of carrier free 109Cd was added to the suspension and mixed with a magnetic stirrer. Subsamples of between 0.5 to 1.0 mL were collected from suspensions after 1, 10, 30 and 60 min (short-term kinetics) and at 1, 3, 7, 11 and 18 days (long-term kinetics). The suspensions were immediately filtered through a 0.2 mm cellulose acetate membrane filter and estimated for radioactivity. Radioactivity in solution was determined with a liquid scintillation counter (Packard 2500 TR) using Packard Ultima Gold Scintillation liquid with a volume/volume ratio of 1 mL of sample to 5 mL of scintillation liquid.
The initial amount of 109Cd introduced (R) was counted together with the concentration of 109Cd remaining in the soil solution after each sample. With the foregoing, there was no need to correct for radioactive decay. A 2 mL subsample of soil solution was collected and filtered through 0.2 mm cellulose acetate membrane before the addition of the radioisotope and soluble Cd was determined by GFAAS.
Analyses of Data
Short-term IEKs were performed on 30 topsoil samples. Isotopically exchangeable Cd values E (t) were computed using r(1)/R and n along with Cd and total Cd using 3 equations below, which represent short-term predicted E(t) values.
| (1) |
where R is the total amount of radioactivity introduced into the system r (1) and r ( ) are the radioactivity remaining in the solution after 1 min and an infinite exchange time, respectively.
n = parameter describing the rate of disappearance of the radioactive tracer from the solution for time longer than 1 min of exchange.
The ratio r (1)/R is the maximum possible dilution of the isotope and is approximated by the ratio of water soluble Cd to the total soil Cd concentration (Fardeau, 1996; Sinaj et al., 1999). It is given as follows:
| (2) |
| where Ccd | = | Total water-soluble Cd (mg Cd L-1) |
| CdT | = | Total Cd digested in concentrated HNO3 and H2O2 (mg kg-1) |
| 10 | = | Factor arising from ratio of 1:10 so that 10 x Cd is equivalent to the water-soluble Cd in soil (mg kg-1) |
Isotopically exchangeable Cd, E(t), was calculated thus:
| (3) |
The above is on the assumption that the soil system is in a steady state.
Statistics
Variation among soils from farmers were estimated using coefficients of variation (CV%) and values were ranked according to the procedure of Aweto (1982) while correlation analysis was performed to relate kinetic parameters with some soil properties.
RESULTS AND DISCUSSION
Soil Properties
Results from the analysis of soil properties are presented in Table 1 with sand-sized fractions dominating in particle size analysis. Textural classes varied from sand to sandy loam. Soils were strongly acidic (pH = 3.7-5.1) but of low organic carbon content (29.8-58.2 kg-1) as well as low cation exchange capacity (2.5-4.6 cmol kg-1).
Surficial Distribution of Cadmium
Total Cd ranged from 12.22 mg kg-1 in farm E to 21.25 mg kg-1 in farm C while exchangeable Cd was least in farm E and highest in farm B (Table 2) slight to moderate variability was shown in exchangeable Cd distribution. The level of Cd in soils of Taiwan ranged from 0.5 to 1 mg Cd kg-1 soil (Chen, 1991; Liu et al., 1998).
Temporal Variability in Kinetic Parameters Using Iek Data
Results of exchange values with time are shown in Table 3. For the 5 soil samples where IEK was conducted for both short term (<60 min) and long-term (18 days) exchange, results indicated that isotopically exchangeable Cd increases with time with relatively large increases occurring between 1 min and 1 day. Thereafter, the rate of increase in exchangeable. Cd decreased and these results were similar to reports by Young et al. (2000) that there was no significant change in the Exchange value (E-Value) in soils of two contaminated mine spoil after 24 h of contact with only a small change occurring after 48 h.
A comparison between measured exchangeable Cd and E(t) values indicated that data from short-term kinetics were successful in predicting exchangeable Cd in two out of 5 soil samples analyzed (farms B and C). When a comparison was made of the r (1)/R between short-term and long-term kinetics, the parameter was significantly greater when estimated for long-term compared with short-term kinetics except for farms C and E. This function was used on the assumption that the total soil Cd, that is the Cd solubilized by concentrated HNO3 and H2O2 digestion is all potentially exchangeable. Studies by Gray et al. (2003) reported that substantial proportions of total Cd are considered non-labile and this may prove the assumption to the contrary.
The ratio of the radioactivity remaining in solution after 1 min of exchange, r (1), to the total introduced radioactivity (r) expressed as r (1)/R ranged from 0.009 for farm E and 0.062 for farm B with an average of 0.043 (Table 4). The rate of decrease in radioactivity for exchange times greater than 1 mm (n-values) ranged from 0.265 to 0.342 (Table 4). These results agree with the findings of Gerard et al. (2000) who reported values between 0.003 and 0.0053 for r(1) / R and n-values ranging from 0.190 to 0.310 for Cd.
| Table 1: | Selected properties of soils studied |
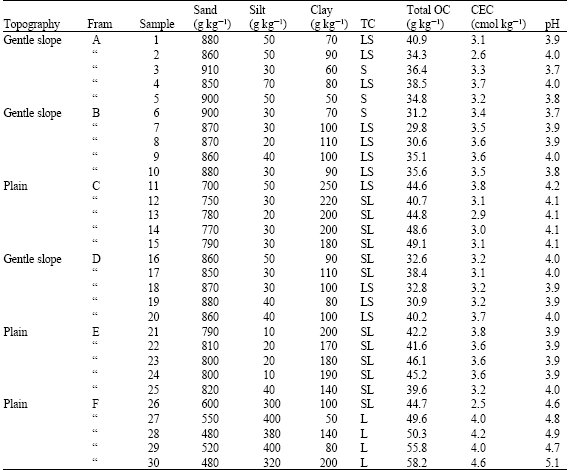 | |
| TC = Textural Class, S = Sand, LS = Loamy Sand, SL = Sandy Loam, L = Loam, Total OC = Total Organic Carbon | |
| Table 2: | Distribution of Cd in the study site (Mean Farm Values) |
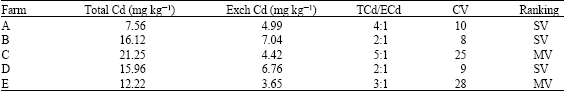 | |
| TCd = Total Cd, ECd, Exchangeable Cd, SV = Slight Variation, MV = Moderate Variation | |
| Table 3: | Nature of kinetic parameters using IEK data between short-term and long-term exchanged |
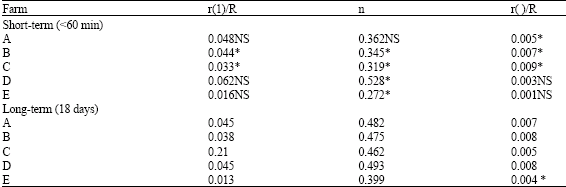 | |
| p<0.05, ns: not significant | |
| Table 4: | Isotope exchange kinetic parameters and isotopically exchangeable Cd (E(t) predicted) for 30 soil samples (mean values of farms) |
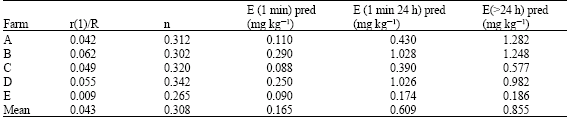 | |
| Pred = Predicted | |
| Table 5: | Correlations(r) between soil Cd kinetic parameters and selected soil properties |
 | |
| ** p<0.01, * p<0.05, NS: Not Significant | |
Relationship Between Soil Cd Kinetic Parameters and Some Soil Properties
There was significant positive relationship between r(1)/R and exchangeable soil Cd and this same trend was followed in the interaction between r(1)/R and total soil Cd (Table 5). There were no significant relations between soil pH and kinetic parameters (r(r)/and N. This statistical result does not imply that pH has no influence in soil Cd concentration. Schwartz et al. (2003) reported a significantly greater Ccd (total water-soluble cadmium) and E (t) values in acidic soils when compared with soils whose lime-amendments caused increase in soil pH. Earlier, a study carried out in Taiwan showed a significant correlation between soil Cd concentration extracted by 0.05 M EDTA or 0.005 M DTPA (p<0.01) and soil pH (Lee, 1999). However, the rate of disappearance of radioactive tracer from the soil of solution for time longer than 1 min of exchange increased as clay content decreased (r = - 0.7; p<0.01). Lee (1999) remarked that clay content and soil pH influence Cd activity and uptake by rice.
CONCLUSIONS
Distribution of Cd in arable soils can be estimated using IEK technique despite local variabilities in soil properties. The IEK technique is helpful in predicting intensity of Cd availability (Ccd) and quantum of occurrence E (t) and these are indicators of how Cd is sorbed or desorbed.
Isotopically exchangeable Cd increased with time with relatively large increases occurring between 1 min and 1 day after which rate of increase in exchangeable Cd decreased. Although soil pH is a key factor affecting solubility and availability, the study has shown that other factors may contribute to Cd availability in soils. Further studies on the compartmental analysis may be necessary to estimate variability in distribution of exchangeable Cd as these farms may have historically received wastes from different automobile vehicles.
ACKNOWLEDGMENT
I appreciate the assistance from Staff, Department of Soil Science, University of Nigeria Nsukka Nigeria.
REFERENCES
- Berti, W.R. and L.W. Jacobes, 1996. Chemistry and phytotoxicity of soil trace elements from repeated sewage sludge applications. J. Environ. Qual., 25: 1025-1032.
Direct Link - Chen, Z.S., 1991. Cadmium and lead contamination of soils near plastic stabilizing materials producing plants in northern Taiwan. Water Air Soil Pollut., 57-58: 745-754.
Direct Link - Ciba, J., T. Korolewicz and M.Truck, 1999. The occurrence of metals in composted municipal wastes and their removal. Water Air Soil Pollut., 3: 159-170.
Direct Link - Fardeau, J.C., 1995. Dynamics of phosphate in soils. An isotopic outlook. Fert. Res., 45: 91-100.
Direct Link - Frossard, E. and S. Sinaj, 1997. The isotope exchange kinetic technique: A method to describe the availability of inorganic nutrients. Applications to K, P, S and Zn. Isotopes Environ. Health Stud., 33: 61-78.
Direct Link - Gerard, E., G. Echevaria, T. Sterckeman and J.L. Moral, 2000. Cadmium availability in three plant species varying in cadmium accumulation pattern. J. Environ. Qual., 29: 1117-1123.
Direct Link - Gray, C.W., R.G. Mclaren and J. Shiowatana, 2003. The determination of labile cadmium in some biosolids-amended soils by isotope dilution mass spectrometry. Aust. J. Soil Sci., 41: 589-597.
Direct Link - Gray, C.W., R.G., Mclaren, D. Gunther and S. Sinaj, 2004. An assessment of cadmium availability in cadmium-contaminated soils using isotope exchange kinetic. Soil Sci. Soc. Am. J., 68: 1210-1217.
Direct Link - Kovacs, B., J. Prokisch, Z. Gyori, A.B. Kovacs and A.J. Palenesar, 2000. Studies on soil sample preparation for inductively couple plasma atomic emission spectrometry analysis. Commun. Soil Sci. Plant Anal., 31: 1949-1963.
Direct Link - Schwartz, C., G. Echevarria and J.L. Morel, 2003. Phytoextraction of cadmium with Thlaspi caerulescens. Plant Soil, 249: 27-35.
Direct Link - Sinaj, S., F. Machler and E. Frossard, 1999. Assessment of isotopically exchangeable zinc in polluted and non-polluted soils. Soil Sci Soc Am. J., 63: 1618-1625.
Direct Link - Young, S.D., A. Tye, A. Carstensen, L. Rescende and N. Crout, 2000. Methods for determining labile cadmium and zinc in soil. Eur. J. Soil Sci., 51: 129-136.
Direct Link








