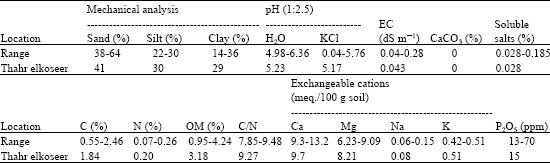
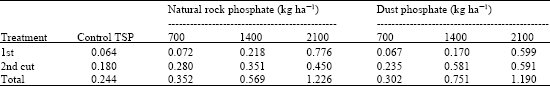
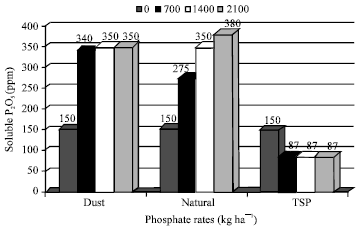
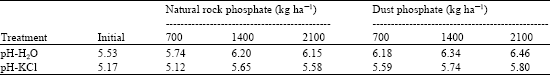Research Article
Release of P from Natural Syrian Rock Phosphate in Acidic Soils
Faculty of Agriculture, University of Damascus, Damascus, Syria
A. I. Rezk
Department of Feftilization Technology, National Research Centre, Cairo-Dokki, Egypt
Mohamed M. El- Fouly
Department of Feftilization Technology, National Research Centre, Cairo-Dokki, Egypt