Research Article
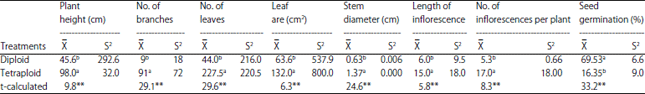
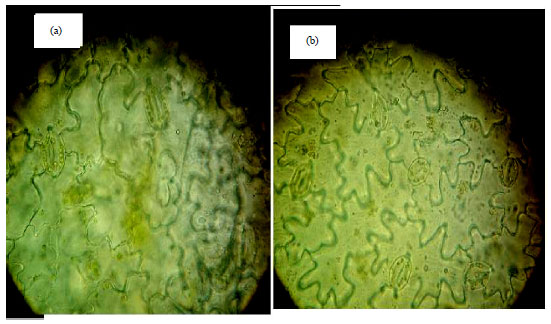
Detection and Evaluation the Tetraploid Plants of Celosia argentea Induced by Colchicines
Department of Horticulture (Ornamental Plants), Faculty of Agriculture, Beni Suef University, Egypt
Mona F. Abou Alhamd
Department of Botany, Faculty of Science, South Valley University, Qena, Egypt