Research Article
Tolerance of Fifteen Soybean Germplasm to Low pH Condition
Indonesian Legume and Tuber Crops Research Institute, Indonesian Agency for Agricultural Research and Development, Jl. Raya Kendalpayak Km 8, Malang, Indonesia
Acidity is common in the world, where 30-40% of the world total area covered by acidity (Haug and Foy, 1984; Von Uexkull and Mutert, 1995). In Indonesia, approximately 69% of dryland faces acidity (Mulyani, 2006). In acid soil, the concentration of H+ is higher than concentration of OH-. The development of soil reaction toward acid or base is supported by certain compounds. Inorganic and organic acids produced by the decomposition of soil organic matter are some of soil constituents that affect soil acidity. Water is also another source of a small amount of H+ ions. Tan (1991) stated that most of the H+ ions present in the soil will be adsorb in complex clay as exchangeable H+ ions. The H+ ions dissociate into free H+ ions. Soil acidity level is determined by the degree of ionization and dissociation in the soil solution. Exchangeable H+ ion is a cause of the formation of potential soil acidity, while the active acidity supported by free H+ ions.
The acidity leads macronutrient deficiencies such as calcium, magnesium, molybdenum and phosphorus, micronutrient toxicities such as aluminum and manganese (Horst, 2000; Thomas et al., 2000). Further, low pH tolerance often coexists with aluminum toxicity and low phosphorus (Liang et al., 2013). This problem in acid soil limits crop growth and development that adversely affect crop production. Lime application is able to diminish acidity effect by increasing the pH that leads decreasing macronutrients deficiency and micronutrients toxicity. Unfortunately, lime application effect takes place for short term period. After the lime reaction disappears the soil properties will be back to the previous acidic soil condition.
Cultivating acidic-tolerant plant can improve plant production in acid soil. This plant has mechanism to decrease acidity effect that allows increasing plant production. Some acid tolerance mechanisms have been reported by some authors. Usually, acidic-tolerant plants have a strong organic acid buffer system in the cell. Foy and Fleming (1978) stated that some acid-tolerant plant such as barley, rice and corn can increase the pH of the nutrient solution in which they are grown. Kuswantoro (2014) also reported different increasing pH levels in the rhizosphere in various seedling ages of six soybean genotypes. Cation Exchange Capacity (CEC) of the soil will decrease in line with decreasing soil pH. According to Howeler (1991), plants also vary on the CEC of the roots. The CEC of the root of acid-tolerant plant is lower than the sensitive plant (Foy, 1984).
In a breeding program, gene source is very important for developing a new superior variety. A gene source can be identified by using the target environment. It is because there is variability in plant species and cultivar within species. Under diverse agro-ecological conditions, phenotypic performance of a genotype is not similar (Ali et al., 2003), because gene expression is subject to modification by the environment (Kang, 1997). Plant species have different tolerance to soil acidity (Hawkins and Robbins, 2010; Phengsouvana et al., 2009) as well as cultivars within species (Foy et al., 1992, 1993). The different tolerance can be identified from the character alteration. One of the major problems in acid soil is acidity beside nutrient deficiency and toxicity. Therefore, acidity can be used as environment target to find out soybean germplasm that tolerance to low pH by studying the character alteration in low pH.
MATERIALS AND METHOD
Experimental design and data collection: The research was carried out in Seed Laboratory of Indonesian Legume and Tuber Crops Research Institute, Malang, Indonesia. The design was factorial design of Randomized Complete Block Design (RCBD). The first factor was pH level containing two treatments of (1) Aquadest with pH 7 as control and (2) Aquadest with pH 4, while the second factor was soybean germplasm consisting 15 accessions of soybean germplasm. Soybean seeds were germinated in a petridish, where each petridish contained 25 sterilized soybean seeds. To ensure the germinated seeds remain standing straight up, a gauze was put inside the petridish. Seed respiration is important in germination to keep the seed life. Therefore, solution was pour up to half of seed size in the petridish. The volume of the solution was monitored everyday and solution was added up to the specified limit when solution decreased due to the absorption by the seeds. Room temperature for germination was maintained at 25°C. The observation was conducted at 6 days after germinating on root length, number of lateral roots, root dry weight, hypocotyl length, epicotyl length, shoot dry weight and seedling dry weight.
Tolerance assessment: The tolerance of soybean genotypes was calculated by using Acid Soil Adaptation Index (ASAI) according to Howeler (1991) as follows:
ASAI = (Ys.Yp)/(s . p)
Where:| ASAI | = | Acid soil adaptation index |
| Ys | = | Yield in stress condition |
| Yp | = | Yield in potential condition |
| s | = | Average yield in stress condition |
| p | = | Average yield in potential condition |
In selecting good varieties, eco-physiological parameters are also important (Zhu et al., 2002) as well as genetic parameters. In acid soil, pH is a critical parameter that influences the physiology of the roots (Hinsinger et al., 2003), where it is expressed in root alteration including root length. Response of the tested genotypes on root length seedling varied between control and low pH and among the genotypes (Fig. 1). Genotype MLGG 0064 showed the highest root length in control condition, while the lowest root length was shown by MLGG 0377. Genotype MLGG 0064 was also showed the highest root length together with genotypes of MLGG 0471, MLGG 0464 and MLGG 0343 in low pH condition, while the lowest were shown by MLGG 0377 and MLGG 0328. The highest decrease percentage on root length was shown by MLGG 0315, MLGG 0304 and MLGG 0064, i.e. 13.14, 12.92 and 11.57%, respectively. Interestingly, there were three genotypes had negative value on decrease percentage, meaning the three genotypes increased in root length (Table 1). It indicates that these three genotypes were the tolerant genotypes. The tolerant genotype can maintain root length under stress conditions (Haling et al., 2011). Genotype background affect dominantly as reported by Kidd and Proctor (2001) that found elongation shoot of Holcus lanatus originating from Flanders Moss but decreasing shoot of H. lanatus originating from Kippenrait Glenn in low pH.
| Table 1: | Decrease percentage of roots length, number of roots and root dry weight of soybean germplasm in Mn toxicity condition |
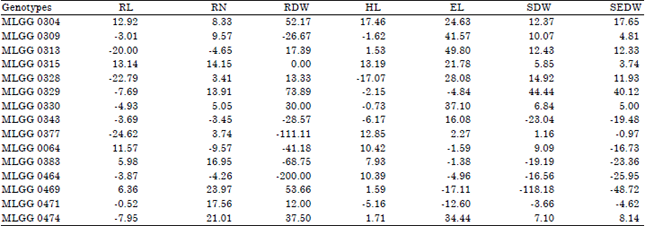 | |
RL: Root length, RN: Root number, RDW: Root dry weight, HL: Hypocotyl length, EL: Epicotyl length, SDW: Shoot dry weight, SEDW: Seedling dry weight | |
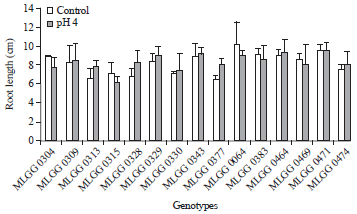 | |
| Fig. 1: | Root length of soybean germplasm in control and low pH conditions |
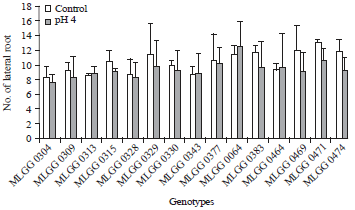 | |
| Fig. 2: | Number of lateral roots of soybean germplasm in control and low pH conditions |
However, different results were reported by Ferrufino et al. (2000) that stated total root length increased 425 fold as the pH was increased from 3.0-6.0. Murata et al. (2003) reported that increasing root length in acid soil was associated with Ca concentration.
Lateral root is more sensitive than primary root (Haling et al., 2011) and low pH stress causes irreversible damage (Koyama et al., 2001). Therefore, this parameter is very important, because the tolerant genotype can be identified from this parameter alteration. The highest number of lateral roots was shown by MLGG 0471 in control condition followed by MLGG 0064 in low pH condition. Generally, most of the tested genotypes showed decrease number of lateral roots in low pH condition except MLGG 0064, MLGG 0464, MLGG 0343 and MLGG 0313 (Fig. 2). Decrease percentages on number of lateral roots of those four genotypes were negative (Table 1). It means that the four genotypes increase in number of lateral roots. Presumably, there was any other factor except genetic factor that increased or maintained number of lateral roots, such as Ca concentration (Murata et al., 2003) or P concentration (Hinsinger et al., 2003). However, the increases in three genotypes were not high reaching 3.45, 4.26 and 4.65% for MLGG 0343, MLGG 0464 and MLGG 0313, respectively. Only MLGG 0064 could reach relatively high increase percentage up to 9.57%. On the other side, the decrease percentages were quite high up to 23.97 and 21.01% for MLGG 0469 and MLGG 0474, respectively. Number of lateral root is initiated by lateral root formation.
Root dry weight is the result of root growth and development including root length and number of roots. Root hair length and root hair density is affected by soil acidity (Haling et al., 2011) lead root dry weight alteration. In control condition MLGG 0469 showed the highest root dry weight followed by MLGG 0471, while the lowest root dry weight were shown by MLGG 0377 and MLGG 0464. In low pH condition the highest root dry weight were shown by MLGG 0464, MLGG 0383 and MLGG 0064 (Fig. 3). There were seven genotypes that experienced increasing root dry weight in low pH condition, while the eight genotypes decreased root dry weight. The previous three genotypes were included in seven genotypes that showed increasing root dry weight in low pH condition (Table 1). In addition, MLGG 0464 also showed the highest increase percentage up to 200%. Other genotype with high increase percentage was MLGG 0377 (111.11%). It indicates that there were different responses among the genotype to low pH condition. Lin et al. (2012) also reported similar data where there was no significant different between pH 6.8 and pH 4 on root dry weight. Similarly, Kuswantoro (2015) reported increasing root dry weight of some soybean genotypes in pH 4 with 75 ppm Mn. Normal root growth is one of the criteria of the tolerant genotypes in low pH except yield growth (Uguru et al., 2012).
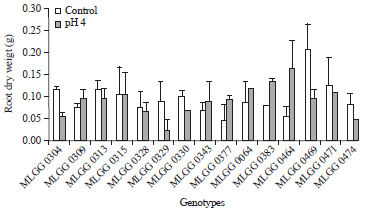 | |
| Fig. 3: | Root dry weight of soybean germplasm in control and low pH conditions |
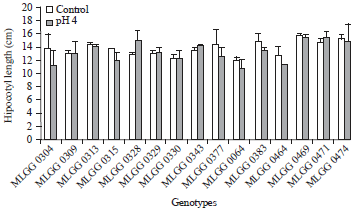 | |
| Fig. 4: | Hypocotyl length of soybean germplasm in control and low pH conditions |
Hypocotyl length of the tested genotypes in control and low pH condition were statistically not different (Fig. 4). However, there were some genotypes that showed relatively high decrease on increase in low pH, such as MLGG 0304 and MLGG 0328 that decreased 17.46% and increased 17.07%, respectively (Table 1). In pH 4 with 75 ppm Mn, Kuswantoro (2015) reported that hypocotyl length in control condition was higher.
Generally, epicotyl length of the tested genotypes in control and low pH condition were lower than in control condition (Fig. 5). However, the highest epicotyl length was achieved by MLGG 0471 in low pH condition. It means that low pH could increase root length in some soybean genotype. There was another genotype having higher epicotyl length, namely MLGG 0469. The increase percentage of MLGG 0471 and MLGG 0469 were 12.60 and 17.11%, respectively. Epicotyl length in pH 4 with 75 ppm Mn was higher than in control condition (Kuswantoro, 2015). Kidd and Proctor (2001) also reported elongation shoot of H. lanatus originating from Flanders Moss but decreasing shoot H. lanatus originating from Kippenrait Glenn in low pH. It indicates that genetics background responded differentially to the low pH. The genotypes with consistent response can be used as gen sources for development a new variety.
Shoot dry weight is the result of shoot growth and development including epicotyl and hypocotyl lengths. The highest shoot dry weight was shown by MLGG 0343 in low pH condition followed by MLGG 0329 in control condition (Fig. 6). The lowest shoot dry weight in control condition was shown by MLGG 0469, while in the low pH was shown by MLGG 0329.
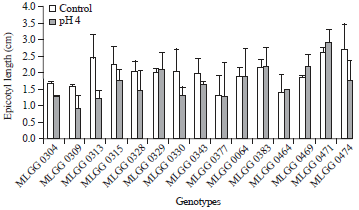 | |
| Fig. 5: | Epicotyl length of soybean germplasm in control and low pH conditions |
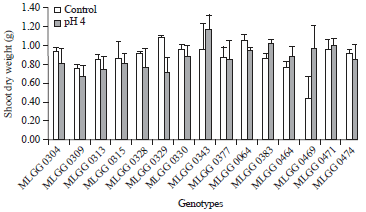 | |
| Fig. 6: | Shoot dry weight of soybean germplasm in control and low pH conditions |
Unlike Murata et al. (2003), who reported decreasing shoot dry weight with decreasing solution pH and Ca concentration, there were five genotypes having shoots dry weight in low pH higher than in control condition. Similar result is reported by Lin et al. (2012) that there was no significant different on shoot dry weight in pH 6.8 and pH 4. The increase percentage of the five genotypes varied from 3.66-118.18% (Table 1). On the other hand, decrease percentage in low pH were not extremely high as the increase percentage, where the highest increase percentage was 44.44% that shown by MLGG 0329. Foy et al. (1993) classified the tolerant genotype by using relative and absolute shoot dry weight. Absolute dry weight is calculated from pH 4, while the relative calculated from pH 4/5.1. In seedlings, the high acidity reduced root growth more than shoot growth (Ritchey and Carter Jr., 1993), because root growth was more adversely affected by the low pH. It demonstrated in hypocotyl length, as one of shoot growth components, that was not significantly different.
The highest seedling dry weight was shown by MLGG 0064 in low pH condition and the lowest was shown by MLGG 0329 also in low pH condition. Genotype MLGG 0329 also showed the highest decrease percentage in low pH reaching 40.12%, whilst the highest increase percentage was shown by MLGG 0469 48.72% (Table 1). Seedling dry weight is the result of root and shoot growth and development including root dry weight and shoot dry weight. The alteration in these two characters affects seedling dry weight alteration. However, for selection criteria in low pH, root dry weight is better than shoot dry weight or seedling dry weight, because root faces directly to low pH condition.
Differential response of the tested genotype indicated different tolerance of the genotypes. Therefore, there were decrease and increase percentage from control to low pH condition.
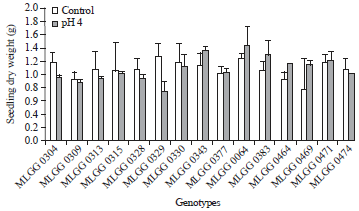 | |
| Fig. 7: | Seedling dry weight of soybean germplasm in control and low pH conditions |
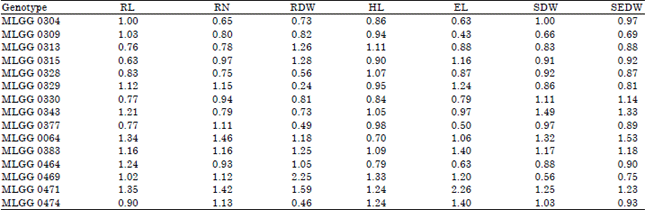
| Table 2: | Acid soil tolerance index of soybean germplasm in low pH toxicity condition |
 | |
RL: Root length, RN: Root number, RDW: Root dry weight, HL: Hypocotyl length, EL: Epicotyl length, SDW: Shoot dry weight, SEDW: Seedling dry weight | |
However, it is difficult to assess the tolerance of the genotypes through the response and decrease-increase percentage of the genotypes. Some researchers had proposed some tolerance or adaptation index such as Tolerance Index (TI) (Khandakar, 1992) and Stress Tolerance Index (STI) (Fernandez, 1993). In this study, the tolerance of the genotypes was assessed by using Acid Soil Adaptation Index (ASAI) (Howeler, 1991). The three highest ASAI values from each character were used as selection criteria for choosing the best genotypes.
Based on the ASAI, there was one genotype having the highest ASAI on all of observed characters, namely MLGG 0471 (Table 2). In potential and stress conditions, this genotype had low different values and high values of all observed characters (Fig. 1-7). The MLGG 0471 also had low decrease percentages on all of characters except number of lateral roots and root dry weight (Table 2). However, these two decrease percentages could be balanced by the high value of the both characters (Fig. 2-3). Beside MLGG 0471, there was other one genotype with the highest ASAI on four characters, namely MLGG 0064. Similarly, this genotype had high values on the four characters (Fig. 1-7). Actually, even though the ASAI on root dry weight and epicotyl length were not included in the highest three genotypes but the ASAI values were remain more than one. It indicates that MLGG 0064 was higher than average of the tested genotypes.
CONCLUSION
The fifteen genotypes responded differently to pH treatments. The observed characters of the sensitive genotypes decreased, while the tolerant genotypes could remain stable or increased. Based on the ASAI, MLGG 0471 was the only genotype that had the highest ASAI on all of the observed seven characters. This genotype can be used as gene source in development soybean variety for low pH tolerance. After MLGG 0471, there was MLGG 0064 that can be considered as a gene source, because it had the highest ASAI on four characters.