Research Article
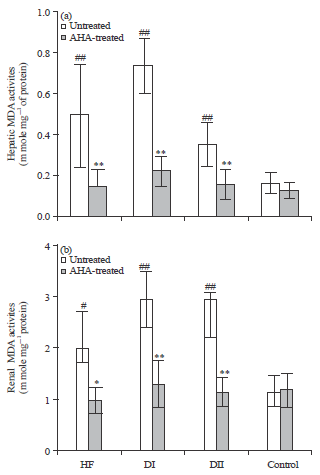
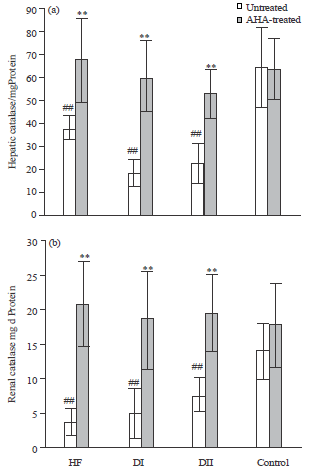
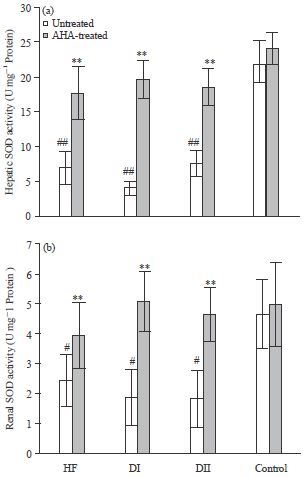
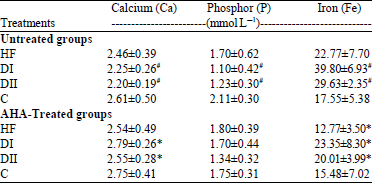
Antidiabetic, Antihyperlipidemic and Antioxidant Effects of Artemisia herba alba Extract on Experimental Diabetes
UR Nutrition, Regulation of Metabolic Systems and Atherosclerosis, High School of Sciences and Technology of Health of Tunis, 176 Bab Souika, BP1006, Tunis, Tunisia
Sarra Khlifi
UR Nutrition, Regulation of Metabolic Systems and Atherosclerosis, High School of Sciences and Technology of Health of Tunis, 176 Bab Souika, BP1006, Tunis, Tunisia
Halima Ben Hmed
UR Nutrition, Regulation of Metabolic Systems and Atherosclerosis, High School of Sciences and Technology of Health of Tunis, 176 Bab Souika, BP1006, Tunis, Tunisia
Inchirah Karmous
UR Nutrition, Regulation of Metabolic Systems and Atherosclerosis, High School of Sciences and Technology of Health of Tunis, 176 Bab Souika, BP1006, Tunis, Tunisia
Anis Benzarti
UR Nutrition, Regulation of Metabolic Systems and Atherosclerosis, High School of Sciences and Technology of Health of Tunis, 176 Bab Souika, BP1006, Tunis, Tunisia
Jalila Elati
Laboratory SURVEN, National Institute of Nutrition and Food Technology of Tunis, 176 Bab Souika, BP1006, Tunis, Tunisia
Abdallah Aouidet
UR Nutrition, Regulation of Metabolic Systems and Atherosclerosis, High School of Sciences and Technology of Health of Tunis, 176 Bab Souika, BP1006, Tunis, Tunisia