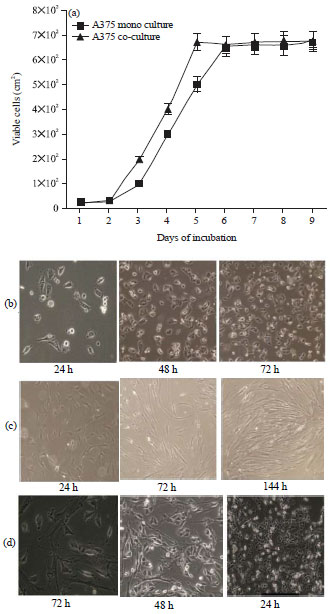
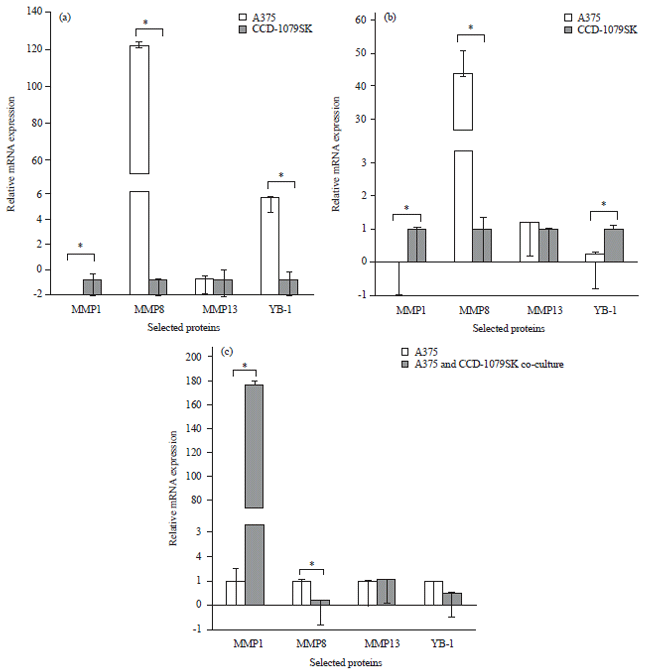
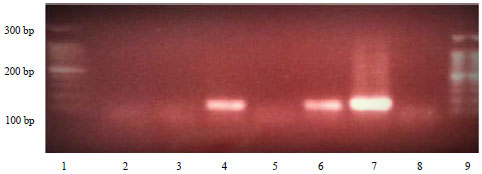
Research Article
Expression of Collagenases Matrix Metalloproteinases and YB-1 Oncogenic Factor in Malignant Melanoma Cancer Cells and its Regulation by Stromal Fibroblasts
Department of Basic Medical Sciences for Nursing, Faculty of Nursing, International Islamic University Malaysia, Kuantan, Pahang, Malaysia
LiveDNA: 964.16203
Ridhwan Abdul Wahab
Department of Biomedical Sciences, Faculty of Allied Health Sciences, International Islamic University Malaysia, Kuantan, Pahang, Malaysia
Mohammad Syaiful Bahari Bin Abdull Rasad
Department of Biomedical Sciences, Faculty of Allied Health Sciences, International Islamic University Malaysia, Kuantan, Pahang, Malaysia