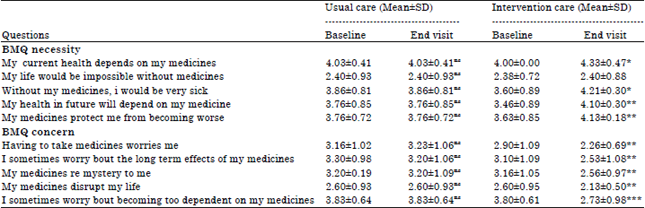
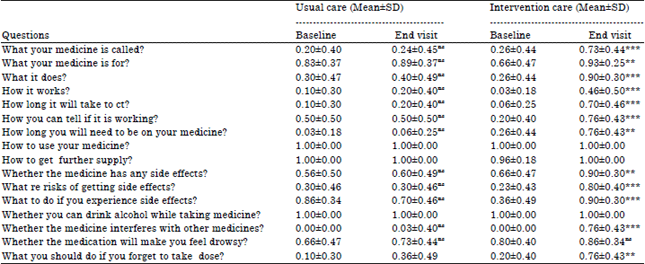
Research Article
Impact of Patient Counseling on Medication Adherence, Beliefs and Satisfaction about Oral Chemotherapies in Patients with Metastatic Cancer at a Super Specialty Hospital
Department of Medical Oncology, Sri Ramachandra University, Porur, Chennai, Tamil Nadu, India
M.G. Rajanandh
Department of Pharmacy Practice, Faculty of Pharmacy, Sri Ramachandra University, Porur, Chennai, Tamil Nadu, India
S. Thanmayee
Faculty of Pharmacy, Sri Ramachandra University, Porur, Chennai, Tamil Nadu, India
G. SalaghaMerin
Faculty of Pharmacy, Sri Ramachandra University, Porur, Chennai, Tamil Nadu, India
S. Suresh
Department of Medical Oncology, Sri Ramachandra University, Porur, Chennai, Tamil Nadu, India
K. Satish Srinivas
Department of Medical Oncology, Sri Ramachandra University, Porur, Chennai, Tamil Nadu, India