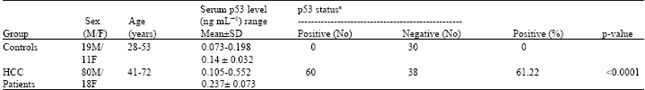
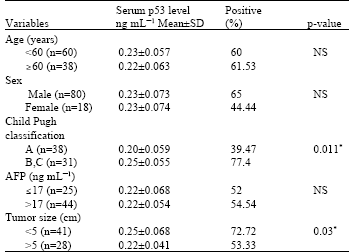
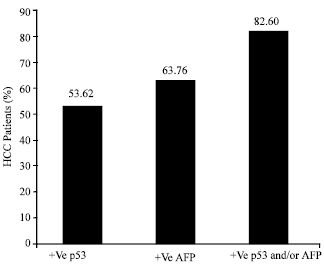
Research Article
Clinical Significance of Serum p53 Antigen in Patients with Hepatocellular Carcinoma
Department of Biotechnology Research, Gastroenterology and Surgery Center, Mansoura University, Mansoura, Egypt
Nabil Ali Gad El-hak
Department of Biotechnology Research, Gastroenterology and Surgery Center, Mansoura University, Mansoura, Egypt
Ayman Tallatt Abbas
Department of Biotechnology Research, Gastroenterology and Surgery Center, Mansoura University, Mansoura, Egypt