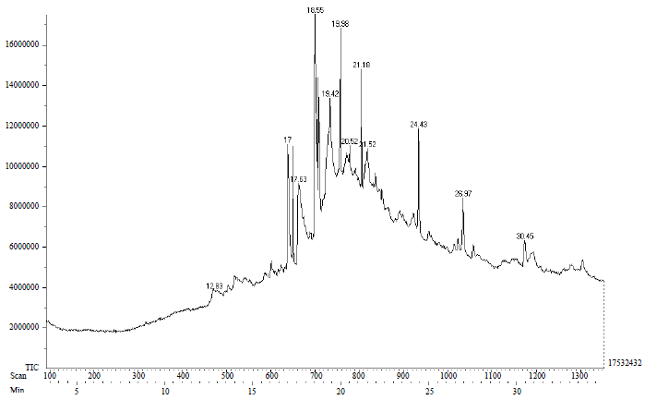
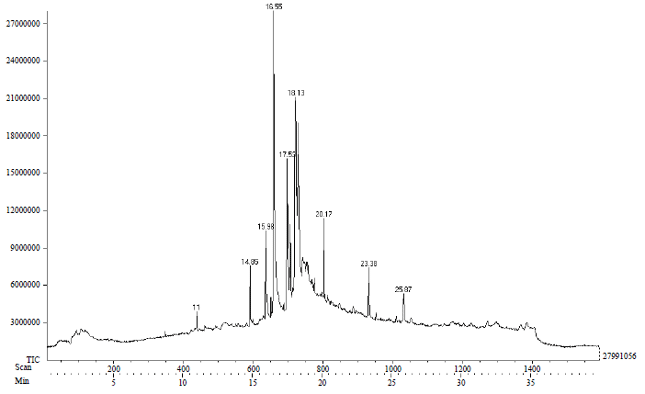
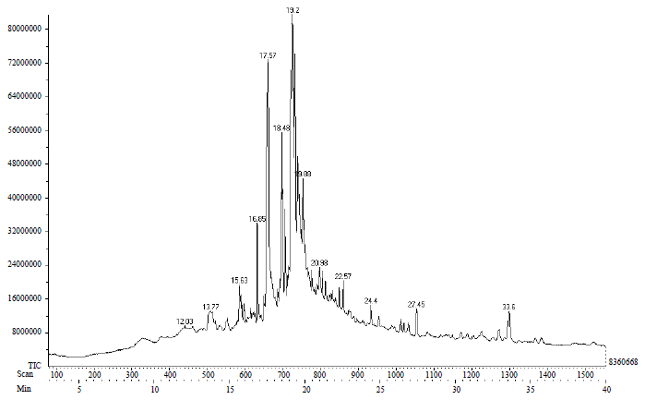
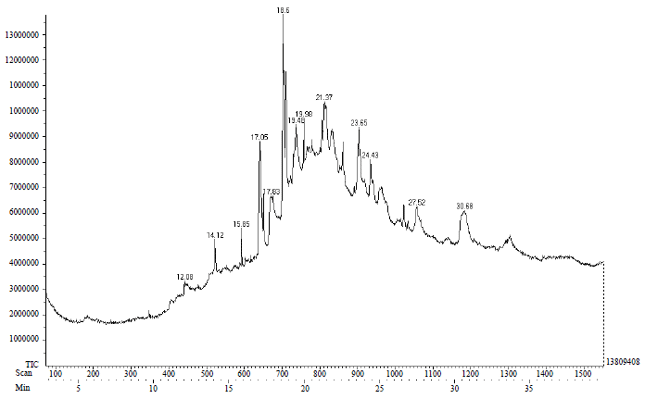
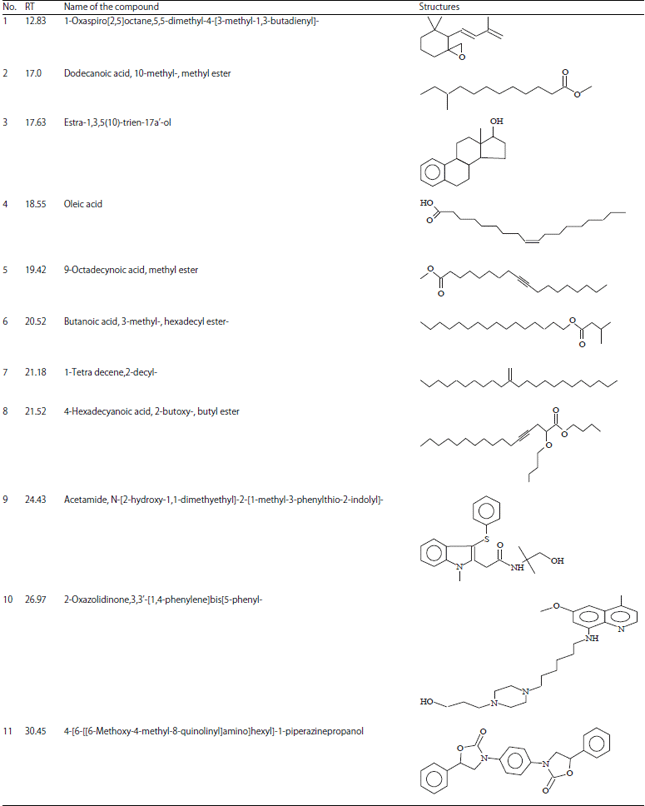
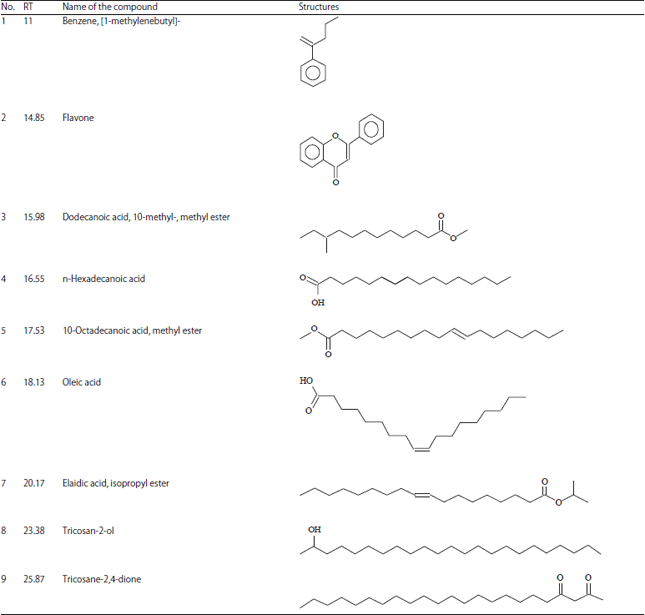
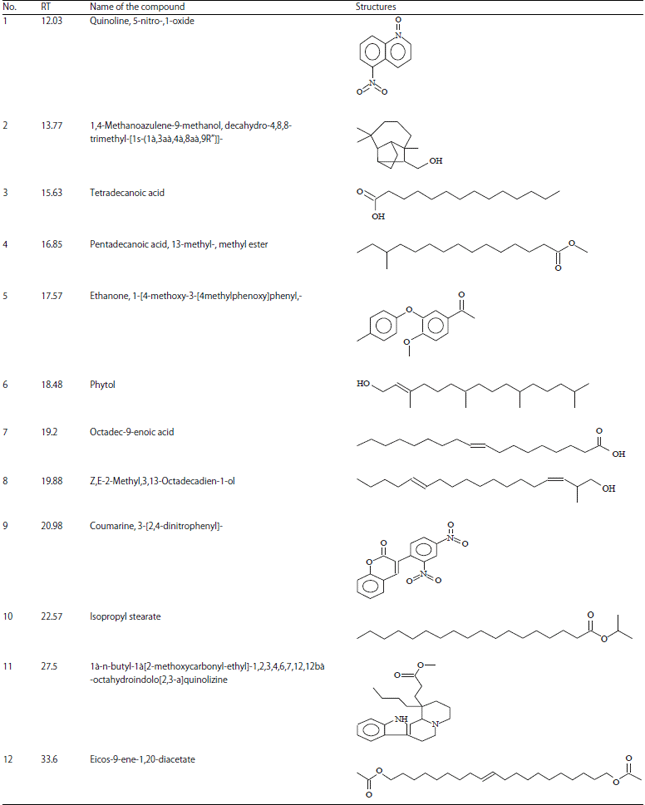
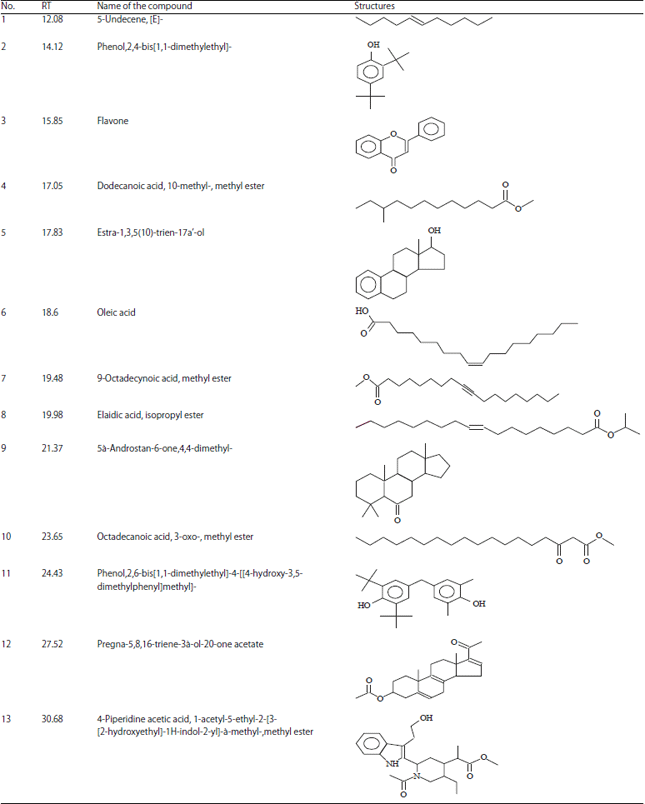
Research Article
Phytochemical Analysis of Flower extracts of Different Cassia Species by Using Gas Chromatography-Mass Spectrometry
Department of Chemistry, Arunai Engineering College, 606603 Tiruvannamalai, Tamil Nadu, India
Shanmugavadivelu Chandra Mohan
Division of Phytochemistry, Shanmuga Centre for Medicinal Plants Research, 613007 Thanjavur, Tamil Nadu, India
LiveDNA: 91.6669