Mini Review
Plant Coumarins as Allelopathic Agents
Department of Biology, Faculty of Sciences, University of Mohaghegh Ardabili, Ardabil, Iran
Allelopathy is defined by the international allelopathy society as any process involving plant or other organisms secondary metabolites that influence the growth and development of surrounding biological systems. This metabolites causing allelopathic effects are called as allelochemicals. The term derives from allochemics coined by Whittaker and Feeny in 1971 and was first used by Chou and Waller in 1983 dealing with interspecific chemical interactions between organisms (Reigosa et al., 2006). Allelochemicals, indeed, are defined as biocommunicatores, suggesting the possibility of active mixtures, because of the increasing number of finding in which single compounds are not active or are not active as active as a mixture (Khalid et al., 2002). Some studies have suggested that allelochemicals may be percent in the mucilage around a germinating seed, in leachates from the aerial parts of plants, in exudates from plant roots, in volatile emissions from growing plants and also among decomposing plant residues where microorganism may also be involved (Naylov, 2002).
In plants allelopathy has study more than other organisms and is regarded as a natural strategy protecting plants against environmental enemies and competing plants. In the last decades, application of synthetic toxins for control of weeds, pests and plant disease caused serious environmental problems. Thus, allelopathy interactions between plants and other organisms may become an alternative to synthetic herbicides and other pesticides (Xuan et al., 2005).
Coumarins are known as a large group pf plant secondary metabolites mainly originated from shikimic acid pathway. This compounds are widely distributed in the Apiaceae, Rutaceae, Asteraceae and Fabaceae families of plants. Coumarins are almost unknown in the animal kingdom and in plant kingdom, this compounds are highly found in 470 most evolved genera of Apiaceae.
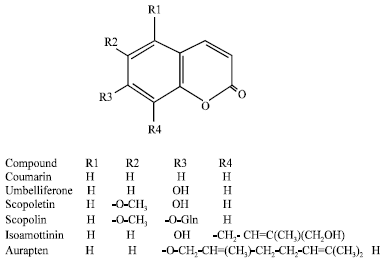 | |
| Fig. 1: | Structure of some allelopathic simple coumarins |
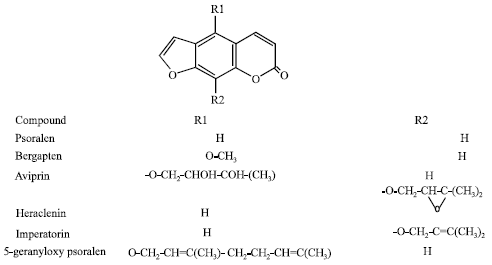 | |
| Fig. 2: | Structure of some allelopathic furanocoumarins |
The compounds are divided into 2 subgroups: simple (Fig. 1 and furano coumarins (Fig. 2). There are a lot of reports on biological activity of both coumarin groups (Rahman, 2000). The diversity of bioactivities among coumarins is so huge that the term pharmacological promiscuity has been applied on their case. These bioactivity also enterprise allelopathic potential of the compounds that can be exploited for enhancing crop production in agroecosystems.
Phytotoxic activity: There are some reports on phytotoxic activity of some Rutaceae plants like Esebeckia yaxhoob and Stauranthus perforatus (Mata et al., 1998; Anya et al., 2005) and Apiaceae species such as Prangos uloptera and Zosima absinthifolia (Razavi et al., 2009a, 2010b). It is assumed that this activity might be attributed to presence of coumarin compounds in the plants. Our previous work revealed that imperatorin, a prenylated furanocoumarin, significantly reduced root and shoot growth of lettuce at concentration higher than 100 μg mL-1. Two simple coumarins, 7-prenyloxy coumarin and auraptene entirely stunt seed germination, root and shoot growth of lettuce at concentration higher than 100 μg mL-1 (Razavi et al., 2010a). We also showed that aviprin, a oxyprenylated furanocoumarin, has indicated phytotoxic activity against lettuce and entirely suppress the seed germination at concentration 500 μg mL-1. However, aviprin glucoside did not exhibited significant phytotoxic activity (Razavi and Zarrini, 2010). Thus, it could be concluded that glycosilition of coumarins reduce their phytotoxic activity. It was also shown that xanthyletin, a pyranocoumarin, indicate very high phytotoxic activity on seed germination and root growth Amaranthus hypochondriacus with IC50 value of 59.9 and 69.5 μg mL-1, respectively (Anya et al., 2005). It was also shown that umbelliferone significantly reduce the growth of some herbs like velvetleaf, pigweed and prosomillet (Shettel and Balke, 1983).
The mechanism of phyto growth inhibitory activity of coumarins was also pointed out. Phytotoxic coumarins inhibit photosynthetic phophorilation in a dose-dependent maner (Anya et al., 2005). The IC50 value for imperatorin inhibitory effects on ATP synthesis in spinach chloroplasts was calculated as 71.5 μg mL-1 (Mata et al., 1998). Coumarin has also been reported to inhibit glycolysis and oxidative phosphoryllation. Xanthotoxin can lower the oxygen uptake by meristematic cells for Allium cepa root tip (Kopidlowasa et al., 1994). Nowadays, in spite of the successful weed control achieved with synthetic herbicides, the use of this chemicals has negative effects on environments and human being, on the other hand, weed species ultimately evolved resistance to a specific herbicide occurred very rapidly (21-3 years) and has led to cross resistance entire chemical classes, underlining the constant need for natural chemicals (Reigosa et al., 2006). Therefore, some coumarins could be candidate for bioherbicides with new target sites.
Antifungal activity: There are numerous reports on the antifungal activity of coumarins. These compounds are inducible antifungal chemicals in plants. When sweet potato roots are attacked by Fusarium oxysporum, some coumarins like umbelliferone, scopoletin and scopolin are produced in the plant tissue. When roots of celery and parsnip were inoculated with Sclerotinia sclerotiorum, a rise in the level of furanocoumarins was observed at the early stage of infection (Rahman, 2000). Therefore, coumarins could be regarded as phytoalexin and may well be considered as a defense tool for plants against pathogenic fungi (Brooker et al., 2007). Some in vitro screening showed that coumarins, especially furanocoumarins, strongly suppress spore germination and mycelia growth of some plant pathogen fungi like Sclerotinia sclerotiorum. We previously showed that imperatorin, a prenylated fouranocoumarin, possess antifungal activity and entirely inhibit mecelia growth of the fungus at a concentration of 1000 μg mL-1 (Razavi et al., 2010b). Psoralen, a unsubstantiated fouranocoumarin, has displayed strong antifungal activity against plant pathogen fungi like Sclerotinia sclerotiorum, Alternaria brassicicola and Cercospora carotae (Al-Barwani and Eltayeb, 2004). Bergapten, 5-methoxy psoralen, was found to have antifungal effects against Alternaria brassicicola, Penicillium expansum and Cercospora petroselini, as well as (Hashem and Saheb, 1999; Al-Barwani and Eltayeb, 2004). It was also shown that ayapin is the most potent antifungal coumarin against Sclerotinia sclerotiorum. In most resistant genotypes of sunflower, ayapin, scopoletin and scopolin were produced in response to pathogen attack in the plant bracts and corollas so when cells accumulate high coumarin concentration. They are exerted to leaf surface to avoid phytotoxicity and to induce resistance against fungal pathogens such as Sclerotinia sclerotiorum and Puccinia graminis (Prats et al., 2007). Two simple coumarins, 5-methyl mullein and 6-methoxy mullein were found to have strong fungitoxic activity against pathogen fungi (Yoshikawa et al., 1979).
The mechanism of antifungal action of coumarins are too complex and far from being completely understood. It was demonstrated that they have induced morphologic changes on the mitochondrial matrix to make it dense. These changes in mitochondrial structure may cause a lack of intracellular energy and so inhibit mitosis (Kopidlowasa et al., 1994).
Coumarins could be utilized to generate a new generation of fungicidal chemicals that are more ecologically friendly.
Insecticidal activity: In sunflower, it was demonstrated that feeding damage of the plant by sunflower beetle, Zygogramma exclamationis induced coumarin biosynthesis. The concentration of ayapin and scopoletin was 3-5 times higher in the damaged plant than control. Induced coumarins subsequently deter additional feeding of sunflower beetles. (Olson and Roseland, 1991).
Other previous researchs have shown that coumarin and murraxocin exhibited strong insecticidal activity and caused high percentage of mortality on eggs and larva of insects (Reda and El-Banhewy, 1986; Sharma et al., 2006). Coumarin is also regarded as an ovicide agent (Nakajima and Kawazu, 1980). It was also demonstrated that some psoralen derivatives like 8-methoxy, 5-methoxy, 5,8-dimethoxy and 5-geranyloxy psoralen were regarded as insect antifeedant agents (Stevewnson et al., 2003). Thus, the development of crop plants using genetic engineering producing coumarins may be a novel approach to control of herbivorous insects.
Antibacterial activity: Up to now, there has been a few works on antibacterial activity of coumarins on plant pathogen bacteria strains, although it was well known that coumarins exhibited high antibacterial effects on animal pathogen strains (Razavi et al., 2009a,b). We previously shown that Isoarnottinin 4'-glucoside displayed antibacterial activity on Erwinia carotovora, a plant pathogen, with MIC value of 100 μg mL-1 (Razavi, 2007).
Nematocidal activity: Regarding to the results of Wang et al. (2008), furanocoumarins possess nematocidal activity. That finding depicted that 8-geranyloxy psoralen, imperatorin and heraclenin indicated nematocidal activity against Bursaphelenchus xylophilus (Steiner et Buhrer) Nickle with medium lethal concentration (LC50) dose of 188.3, 161.7 and 114.7 mg L-1 at 72 h, respectively. These compounds also showed nematocidal effects against Panegrellus redivivus (Linn.) Goodey with LC50 value of 117.5, 179.0 and 184.7 mg L-1 at the same time course, respectively (Wang et al., 2008).