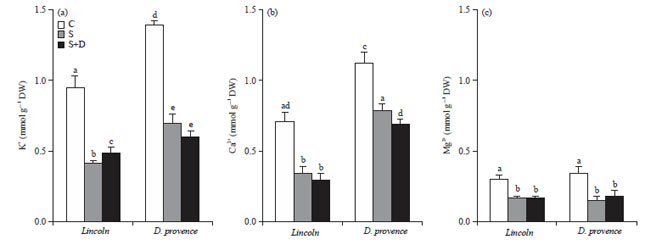
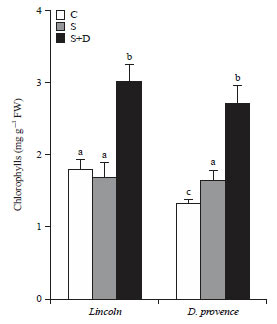
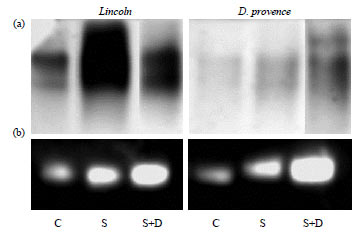
Research Article
Interaction Between Salt Stress and Drought Stress on Some Physiological Parameters in Two Pea Cultivars
Department of Biology, Faculty of Science, Taif University, Taif, Kingdom of Saudi Arabia
LiveDNA: 216.30371
Khalid H Alamer
Department of Biology, Science and Arts College-Rabigh Campus, King Abdulaziz University, Jeddah, Kingdom of Saudi Arabia
Chayma Ouhibi
Physiology and Biochemistry of Plant Response to Abiotic Stress, Faculty of Science of Tunis, Tunis El Manar University, 2092 Tunis, Tunisia
Samia Oueslati
Biotechnology Center of Borj-Cedria, University of Tunis El Manar, Tunis, Tunisia
Mokhtar Lachaal
Physiology and Biochemistry of Plant Response to Abiotic Stress, Faculty of Science of Tunis, Tunis El Manar University, 2092 Tunis, Tunisia