Research Article
Optimization of Plant-Bacteria Complex for Phytoremediation of Contaminated Soils
Department of Plant and Microbial Biology, University of California, Berkeley, 111 Koshland Hall, Berkeley, 94720, California
Soil and water pollution with trace metals is one of the most important environmental problems in industrialized countries by producing harmful consequences for agriculture and human health (Schröder et al., 2007). Current remediation methods applicable to contaminated soils are expensive, environmentally invasive and labor intensive. This problem can be obviated by using far less expensive plant-microbe treatment systems to farm the contaminant out of the soil or sediment. This process is referred to phytoremediation, i.e., the use of plants and their associated microbiota to remediate environmental contaminants. In the rhizosphere, sugars and amino acids may be released directly from roots in the form of exudates or by the decay of fine roots to the microbes growing in the immediate proximity to roots. Thus, plants facilitate organic contaminant reduction principally by providing an optimal environment for microbial proliferation in the root zone (Lee et al., 2001).
The advantage of this farming approach is that it is much more cost-effective than both physicochemical approaches and bioremediation. It can be used to remove pollutants over very large areas. Furthermore, the soil is remediated on site, eliminating transportation costs. In this method, soil bacteria that interact specifically with plant roots in the rhizosphere (rhizobacteria) inducing phytoremediation either by forming symbiotic relationships with the plant or by microbial biotransformation of organic compounds, thereby indirectly facilitate their removal. These degradative processes are influenced not only by rhizosphere microorganisms, but also by unique properties of the host plant (Chaudhry et al., 2005). If plants can be successfully established on polluted soils, then the plant-microbe combinations in the rhizosphere can provide an economical method for enhancing the microbial degradation of complex contaminants.
Several studies have examined introducing contaminant-degrading bacteria (naturally or genetically modified) to enhance the rate of biodegradation in polluted sites (Hanson et al., 1997; Somers et al., 2004; Tesema et al., 2004; Wei et al., 2003). Indian mustard plants inoculated with rhizosphere bacteria had 5 fold higher Se concentration in roots and 4 fold higher rate of Se volatilization compared with axenic controls (de Souza et al., 1999). Certain soil microorganisms have been shown to significantly enhance Zn accumulation in the shoot of the hyperaccumulator Thlaspi caerulescens by facilitating an increase in the solubility of non-labile Zn in the soil and thus enhancing its bioavailability to this plant (Whiting et al., 2001). In a similar manner, root exudates significantly enhanced phenanthrene biodegradation in rhizosphere soils, either by increasing contaminant bioavailability and/or increasing microbial population size and activity (Miya and Firestone, 2001). One difficulty with studying phytoremediation using plant-microbe combinations is that not all components of the contaminated mixture are degraded with equal efficiency. To date, little research has been done on devising methods for phytoremediating a mixture of different metals using multiple bacterial species.
As many variables are involved, experimental designs are powerful tools to optimize a certain procedure. Among the different groups of designs, Plackett-Burman design introduced by Plackett and Burman (1946), allows the main effects of a greater number of factors (k+1) to be known with relatively few experiments (Bermejo-Barrera et al., 2000). The design can be used only when k+1 is a multiple of 4 (i.e., k = 3, 7, 11, …). Although such design provides a simultaneous examination of the entire suite of parameters, it has not been widely used in ecological research (Beres and Hawkins, 2001).
The aim of the present study is to optimize the plant-microbe combinations to clean up multi-metal contaminated soils. Indian mustard plant (Brassica juncea L.) has been used as a target plant. This species has previously been demonstrated to be metal tolerant and an excellent candidate for phytoremediation (Clemens et al., 2002; Pilon-Smits, 2002; Salt et al., 1995). Three different Bacillus bacterial strains (isolated from highly contaminated sediment) were introduced into the rhizosphere of Indian mustard in soil contaminated with mixture of selenium (Se), cadmium (Cd) and chromium (Cr), to study the plant-microbe interactions on metal removal. Because there are many variables that affect the remediation process, the experimental design Plackett-Burman has been used.
Isolation of bacterial strains with multiple heavy metal resistance phenotypes: The experimental bacteria were collected aseptically from different contaminated environments from the area of Marute Lake and Eastern Harbor in Alexandria City, Egypt. Molecular phylogenetic study using 16S-ribosomal RNA sequencing revealed that the bacterial strains are Bacillus thuringiensis (WS 2625), Bacillus licheniformis (NCIMB 6816) and Bacillus biosubtyl.
Experimental design: The Plackett-Burman fractional factorial design was applied to reflect the relative importance of 7 various environmental factors involved in metal removal with only 8 experiments instead of the 27 (128) required for a full factorial design. The design allowed each factor to be examined in 4 trials at a low level (-1) and in 4 trials at a high level (+1). The experimental matrix is shown in Table 1.
| Table 1: | Plackett-Burman experimental matrix for 7 factors |
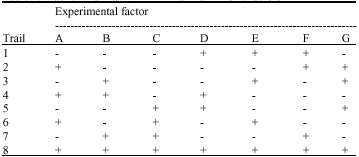 | |
| +: Positive; -: Negative | |
| Table 2: | Screening for the levels of experimental parameters (7-factors) affecting plant-microbe combination for phytoremediation of trace metals based on the results of the Plackett-Burman design shown in Table 1 |
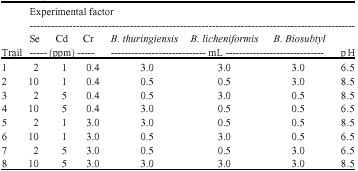 | |
In the present study, three factors are related to the concentration of a specific metals Se, Cd or Cr in the soil. Three other factors corresponded to the inoculation of different bacterial strains, Bacillus thuringiensis (WS 2625), B. licheniformis (NCIMB 6816) and B. biosubtyl. The seventh factor was the soil-pH. The effect of changing each factor from a low to a high level was examined by increasing shoot and root length and biomass and metal accumulation in plant tissues. The factors studied as well as the values in each trail are represented in Table 2. Trials were preformed in triplicate.
The main effect of each variable was calculated as the difference between the average measurements at the high level (+1) and the average measurements at the low level (-1) of that factor. One-way ANOVA was applied to examine the significant differences between the high and low level for each factor (Chatfield, 1983).
Effect of plant-microbe interactions on metal removal: The effects of microbes on heavy metal uptake were investigated using Indian mustard plant (Brassica juncea L.). Seeds were surface sterilized by shaking in 70% ethanol for 30 sec, followed by 10% sodium hypochlorite for 30 min in tightly closed, sterile plastic tubes. Seeds were then washed 5 min washing with sterile Double-Distilled Water (DDW).
The bacterial mixtures were prepared in sterile MOPS buffer with different densities and pH required for each trial. Inocula from fresh slants of the three bacillus strains (Bacillus thuringiensis (WS 2625), B. licheniformis (NCIMB 6816) and B. biosubtyl) were added to 10 mL of LB medium in 100 mL Erlenmeyer flask. The flasks were inoculated from a pre-culture at 37°C on rotary shaker (250 rpm) until late stationary phase (OD550 = 1.0). An overnight seed culture of each Bacillus isolate was prepared by inoculating 0.5 mL of each strain in flasks containing 50 mL of pre-warmed fresh basal medium at 37°C on rotary shaker (250 rpm). To adjust the bacterial density in the medium, 0.5 mL of the seed culture was transferred to 50 mL pre-warmed fresh medium. After 4 h of shaking at 37°C, A550 as measured until it reaches 1.0. At this stage inoculum size of 3 and 0.5 mL were used to represent the high and low density of bacterial cells used in the different trials, respectively.
The metal mixtures were prepared in (DDW) with the concentrations based on mg g-1 dry weight of soil as shown in Table 2. The metal solutions were sterilized using 0.22 μm filter.
Fifty grams of fresh soil (UC-mix) were placed in Magenta boxes (Sigma). The boxes were autoclaved twice for 45 min (dry cycle) to kill the native microbial populations in the soil. Twenty-five surface sterilized seeds of Indian mustard were transferred aseptically into the soil of three replicate Magenta boxes. The multi bacterial strain mixture then was added to each trial. To avoid a temperature stress or low aeration inside the box, boxes containing soil and seeds were closed using other sterile boxes connected by an adapter to increase the air volume inside the boxes. A Nalgene (Milwaukee, WI) tube was inserted through the top of the box and perfectly sealed. The outer end of the tube was connected to a 0.22 μm filter by rubber tube to sterilize the air so that the plants could be aerated. The Magenta boxes were placed in a controlled-environment plant growth chamber with constant light at 25°C and sterile DDW was added aseptically as required.
After 10 days growth period, the specific sterile metal mixture was added to each box. This 10 days growth period was needed to allow the bacterial strains to establish in the rhizosphere. The plants kept growing for an additional 35 days in the same controlled-environment as described earlier.
At harvest, Indian mustard plants were removed from the boxes, rinsed thoroughly in DDW to remove adhering soil and the shoot and root lengths were measured. Soil from each box was air-dried and 1 g sub-samples of this soil were used for chemical analysis. The plants were separated into shoot and root, were dried at 80°C and dry mass was weighted. The concentration of Cd and Cr in plant tissues as well as in the soil were determined by Inductively Coupled Plasma Atomic Emission Spectroscopy (ICP-AES) following digestion in 4 mL of concentrated HNO3 at 85°C for 4 h (Fassel, 1978). Se concentration in plant and soil was carried out using acid digestion (de Souza et al., 1998) followed by hydride generation-atomic absorption spectroscopy (Ward and Gary, 1996). The detection limit of Se in this method was 1 μg L-1. A wheat flour standard (Se at 1.1 mg kg-1) and a blank were used with all digestions. All samples were diluted in 6M HCl to give absorbance in the linear portion of the standard curve.
The influence of different bacterial strains in the rhizosphere of contaminated soil was evaluated by comparing the main effect on metal accumulation in plant tissues as described in the introduction. When the value of main effect of the tested variable is positive, the influence of this variable upon metal accumulation is superior at high level. On the other hand, a negative value of the main effect means that the influence of the variable is superior at a low level.
The results showed that inoculating high-density of B. licheniformis in the rhizosphere of Indian mustard was enhanced Cd accumulation in plant tissues. Whereas, Cd accumulation was no considerably affected by the presence of B. thuringiensis and B. biosubtyl in the rhizosphere as indicated by a negative values of the main effect (Fig. 1). The positive value of the main effect of soil`s pH indicated that the high pH level used is optimum for B. licheniformis activities in the rhizosphere to enhance Cd accumulation in Indian mustard tissues.
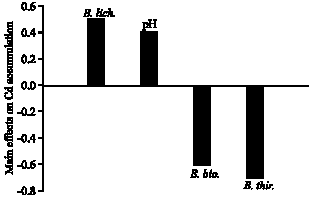 | |
| Fig. 1: | Effect of the bacterial strains Bacillus thuringiensis (WS 2625), B. licheniformis (NCIMB 6816) and B. biosubtyl and pH level on Cd accumulation in Indian mustard plant tissues based on the main effect results of the Plackett-Burman experiment |
Several mechanisms responsible for Cd detoxification in plants include production of phytochelatins (PCS) in cytosol (low molecular weight Cd-PC complexes) or compartmentation in the vacuole (high molecular weight Cd-PC complexes) (Rauser, 1999). Heiss et al. (2003) indicated that Indian mustard treated with 25 μM Cd responded by increasing the PCS in leaves and shoots 3-fold higher than in roots as a defense system for Cd detoxification. Wójcik et al. (2005) reported a minor role of PCS in Cd tolerance in hydroponically-grown Thlaspi caerulescens (Brassicaceae) at concentration range from 5 to 500 μM. In solving these problems it is crucial to learn the genetic mechanisms beyond metal hyperaccumulation to improve the plants for eventual use in phytoremediation.
Inoculating of high densities of examined bacterial populations in the rhizosphere of Indian mustard was assisting on the removal of Cr from the soil as indicated by positive values of the main effects (Fig. 2). B. licheniformis recorded the highest score (+0.93) for accumulating Cr followed by B. biosubtyl (+0.76) and B. thuringiensis (+0.24). However, Cr accumulation was enhanced at low soil-pH (main effect: -0.32). This higher accumulation of Cr in Indian mustard in the present study supports the findings by Zayed et al. (1998) who reported that the members of the Brassicaceae have the ability to accumulate high concentration of Cr in root, while translocation from roots to shoots is extremely limited. Lytle et al. (1998) described the role of rhizosphere bacteria in Cr accumulation in saltmarsh bulrush in which plant actively but rapidly reduced Cr (VI) upon entry into the roots and only accumulated Cr (III) in the roots and shoots.
Accumulation of Se in Indian mustard was substantially higher when the bacteria were inoculated into the rhizosphere at low density (Fig. 3). The highest Se accumulation was achieved in association with B. licheniformis in the rhizosphere (-38.6) and B. thuringiensis (-2.49) followed by B. biosubtyl (+0.65) (Fig. 3). This result may demonstrate the little effect of bacterial population on Se accumulation in plant tissue. Indian mustard is known to be the most efficient in extracting Se from soil and efficiently volatilizes and accumulates Se in its tissues (de Souza et al., 1998; Terry and Zayed, 1998). The kinetic study of de Souza et al. (1998) has shown that selenate uptake and maintenance in the root may be rate limiting for Se assimilation and volatilization by Indian mustard. Furthermore, rhizosphere bacteria can overcome part of the rate limitation, i.e., Se uptake and thereby increase Se accumulation in tissues and volatilization.
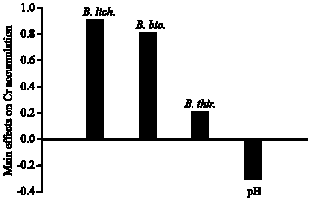 | |
| Fig. 2: | Effect of the bacterial strains Bacillus thuringiensis (WS 2625), B. licheniformis (NCIMB 6816) and B. biosubtyl and pH level on Cr accumulation in Indian mustard plant tissues based on the main effect results of the Plackett-Burman experiment |
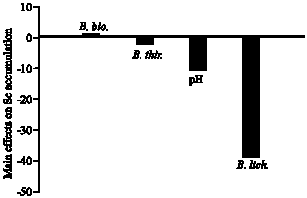 | |
| Fig. 3: | Effect of the bacterial strains Bacillus thuringiensis (WS 2625), B. licheniformis (NCIMB 6816) and B. biosubtyl and pH level on Se accumulation in Indian mustard plant tissues based on the main effect results of the Plackett-Burman experiment |
The interaction between different factors in the soil was though to affect the plant growth parameters, which in turn may influence the metal accumulation in the plant tissues. The present study showed that the plant growth parameters (e.g., shoot and root length and biomass) were enhanced by presence of bacteria in lower densities in the rhizosphere of Indian mustard as indicated by negative values of the main effect (Fig. 4). Rhizosphere bacteria can simulate plant growth in numerous ways such as enhancing mineral and water uptake; producing phytoremediation and altering root morphology (Fallik et al., 1994; Kapulnik, 1996; Lin et al., 1983).
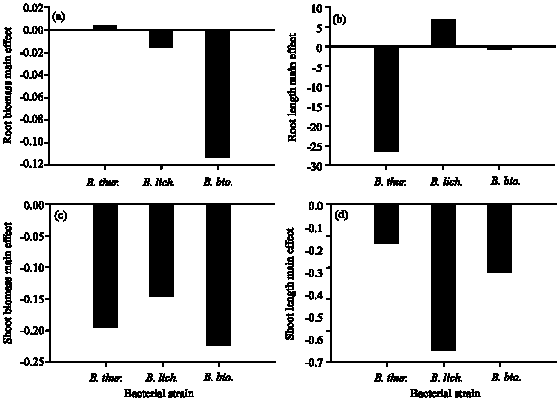 | |
| Fig. 4: | Effect of the bacterial strains Bacillus thuringiensis (WS 2625), B. licheniformis (NCIMB 6816) and B. biosubtyl on growth parameters (shoot-root lengths and biomass) of Indian mustard plant based on the main effect results of the Plackett-Burman experiment |
Therefore, it is unlikely that the simulation of plant growth by bacteria is responsible for enhancing metal accumulation in the plant tissue.
Since contaminated sites in nature usually include a mixture of different contaminants as well as a diversity of microorganisms, one important area in need of more study is the cross-linked trace metals-rhizosphere bacteria complex as a potential system for phytoremediation. Because of the great complexity of the problem, a statistical design, i.e., Plackett-Burman method will be very beneficial for evaluation of the phytoremediation processes because it shows the best conjunction between the bacterial strains with other plant species to improve their phytoremediation potential.
Indian mustard, in one hand, has been identified as a plant species that has a greater ability to absorb, sequester and accumulate different metals, grow rapidly on contaminated soil, tolerate salinity and other toxic conditions and provide a safe source of forage for metal-deficient livestock. Multiple heavy metal resistance bacteria, on other hand, provide promising prospects for future phytoremediation studies. The results from the present study suggest that bacteria that were identified in the laboratory as being superior for enhancing metal phytoextraction could be tested under field conditions. For example, germinating bacteria-coated seeds in metal contaminated soil could make the phytoremediation of contaminated sites more efficient if the selected bacteria can compete favorably with the resident populations. In addition to being an excellent species for metal phytoremediation, Indian mustard is an excellent candidate for the phytoremediation of metals from contaminated soil and water through processes such as phytoextraction and rhizofiltration (Dushenkov et al., 1995). Also, Indian mustard used in conjunction with rhizosphere bacteria that are superior at enhancing plant metal accumulation from contaminated soils and agricultural drainage water.
The author would like to thank Professor Soraya Sabry; Department of Botany; Faculty of Science; Alexandria University, Egypt for kindly providing the bacterial isolates used in this study. The author acknowledges with gratitude the lab facility provided by Professor Norman Terry, University of California, Berkeley to complete this study.