Research Article
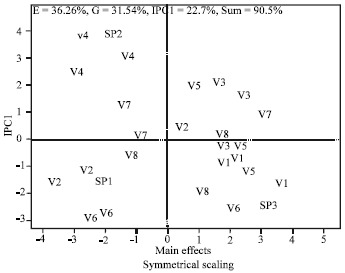
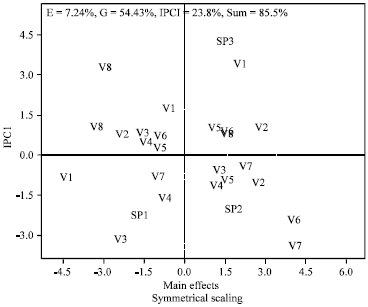
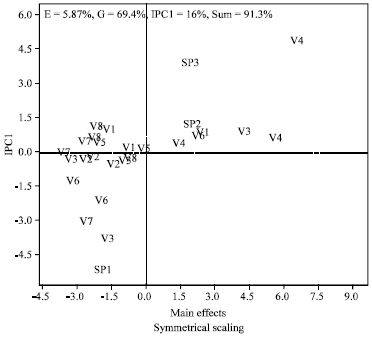
Genotype by Environment Interaction of Chlorophyll Reduction in Rice Cultivars Screened for Resistance to Rice Yellow Mottle Disease with Vector Transmission Method
Joseph Ayo Babalola University, P.M.B. 5006, Ilesa, Osun State, Nigeria
D.B. Olufolaji
Federal University of Technology, P.M.B. 704, Akure, Ondo State, Nigeria
F.E. Nwilene
Africa Rice Center (AfricaRice), P.M.B. 5320, Ibadan, Nigeria
A. Onasanya
Africa Rice Center (AfricaRice), P.O. Box 33581, Dares-Salaam, Tonzania
M.M. Omole
Joseph Ayo Babalola University, P.M.B. 5006, Ilesa, Osun State, Nigeria
R.O. Onasanya
Federal University of Technology, P.M.B. 704, Akure, Ondo State, Nigeria
Y. Sere
Africa Rice Center (AfricaRice), 01 BP 2031, Cotonou, Benin