Short Communication
Combinatorial action of essential oils towards pulse beetle Callosobruchus chinensis Fabricius (Coleoptera: Bruchidae)
Department of Zoology, Mahatma Gandhi Post Graduate College, Gorakhpur-273001, (U.P.), India
The practice of storing food grains began about 4,500 years ago as a safeguard against poor harvests and famines due to adverse weather conditions. Since time immemorial several species of storage insect pest attacked granaries and other food structures and even today storage losses remain notoriously high. According to an estimate, the overall damage caused by stored-grain insect pests accounts for 10-40% worldwide annually. In India, food grain losses during storage at the farm level approximate 10% of the total production. Inspite of improved storage structures and modern chemical and physical control techniques employed for the safe storage of stored grains, 70-90% of food grain is still stored for six months to a year at farmer’s level in traditional storage structures. In such a critical situation, protection of stored grains from insect infestation is quite essential. To achieve this goal, the use of several synthetic pesticides came into existence. But residues of such pesticides accumulate at different trophic levels in food chain and exceed the tolerance limit. Experimental studies of chronic exposure to pesticide have revealed increased risk of neurotoxic, carcinogenic, teratogenic and mutagenic effects among others. Also, many insects have acquired resistance against various synthetic insecticides. In addition, efficacy of these synthetic insecticides against stored-grain insect pests varies greatly after treatment. Uncontrolled use of these synthetic insecticides causes great environmental hazards due to their persistent nature and also various biochemical and behavioral changes in non-target animals (Gupta et al., 2001).
Callosobruchus chinensis is a serious pest of stored grain which infests cowpea, beans, lentils and other pulses. In India, it is responsible for 32-64% loss under storage conditions and maximum damage occurs from April to October (Mukherjee and Joseph, 2000). Only grubs are infective stages for the storage grains. These make holes in the grains and consume the inner part leaving empty kernel. The damaged grains become unsuitable for human consumption, production of sprout and also lose its market value. For the control of these specific and other stored grain insect pests, efforts have been made to develop new alternatives of synthetic pesticides. In such strategies, inert dusts (Mahdi and Khalequzzaman, 2006), ionizing radiations (Valizadegan et al., 2009), ozonization (Hollingsworth and Armstrong, 2005), microbial products (Buda and Peciulyte, 2008; Khashaveh et al., 2008; Mahdneshin et al., 2009), plant extracts (Allotey and Azalekor, 2000; Sabbour, 2003; Upadhyay et al., 2006; Haghtalab et al., 2009), insect growth regulators (Arthur and Hoeremann, 2004) and biological agents have been proved effective tools in controlling stored grain insect pests. From the last two decades, plant products especially essential oils have been gaining much attention in insect pest management programme. Till now, many essential oils and its constituents have been reported for insecticidal activities against stored grain insect pests (Lee et al., 2001; Tripathi et al., 2003; Koul et al., 2007; Chaubey, 2007, 2008; Aboua et al., 2010; Manzoomi et al., 2010). Present investigation aimed to study insecticidal activities of different combinations of T. ammi, A. graveolens and N. sativa essential oils against C. chinensis.
Isolation of oils: The dried fruits of Ajowan (Trachyspermum ammi, Umbelliferae), dill (Anethum graveolens, Umbelliferae) and black cumin (Nigella sativa, Ranunculaceae) were purchased from the local market of Gorakhpur, (U.P.), India. These were grounded by domestic mixer and the powdered material was hydrodistilled in Clevenger apparatus continuously for five hours to yield essential oils. The oils were collected in glass containers and kept at 4°C until their use.
Insects: Pulse beetles C. chinensis was used to determine the insecticidal nature of different combinations of essential oils. The insects were reared on cowpea in laboratory at 30±2°C, 75±5% RH and a photoperiod of 10:14 (L: D) h.
Toxicity assay: Insecticidal properties of T. ammi and A. graveolens; A. graveolens and N. sativa; and N. sativa and T. ammi essential oil combinations were tested against C. chinensis adults by fumigation method. Glass vials (10 cm long and 8 cm diameter) with polystyrene cap were used for this assay. For fumigation, filter paper strips (1 cm2) treated with solutions (prepared in acetone) of different concentrations of different essential oil combinations was used. Ten grams of cow pea grains and ten adults were taken to each vial and the open end of the vial was closed by the cap so that the oil treated filter paper strip remained inside the vial. For each combination of essential oils, four different concentrations and for each concentration of essential oil combination, six replicates were used. In all combinations, different essential oils were mixed in equal proportion. The vials were kept at 30±2°C, 75±5% RH and a photoperiod of 10:14 (L: D) hours and mortality in insects were recorded every after 24 h of treatment up to 96 h. In control, untreated filter paper strip treated with acetone only was used.
Oviposition inhibition assay: Effects of T. ammi and A. graveolens; A. graveolens and N. sativa; and N. sativa and T. ammi essential oil combinations on oviposition potential was studied by fumigating newly emerged adults of C. chinensis with two sublethal concentrations viz. 40 and 80% of 24 h LC50 of the essential oils as was done in toxicity assay. For fumigation, filter paper strips (1 cm2) treated with solutions (prepared in acetone) of different concentrations of different essential oil combinations were used. Ten grams of cow pea grains and ten adults were taken to each vial and the open end of the vial was closed by the cap so that the oil treated filter paper remained inside the vial. For each combination of essential oils, four different concentrations and for each concentration six replicates were used. In all combinations, different essential oils were mixed in equal proportion. The vials were kept at 30±2°C, 75±5% RH and a photoperiod of 10:14 (L: D) h and number of eggs were recorded after 96 h of treatment. In control, untreated filter paper was used. The %ODI was calculated as 100(A-B)/(A+B), where A and B represent the number of eggs laid in control and in the test, respectively.
Data analysis: Median lethal concentration LC50 was calculated by Probit Or LOgit (POLO) programme (Russell et al., 1977). Analysis of variance was performed to determine significancy of the data and dose response relationship (Sokal and Rohlf, 1973).
Fumigation with different combinations of essential oils caused death of C. chinensis adults. The LC50 values of different combinations of T. ammi and A. graveolens; A. graveolens and N. sativa and N. sativa and T. ammi essential oils adults were 11.6, 10.2, 9.6 and 8.5; 8.7, 7.4, 6.8 and 5.9 and 11.2, 10.5, 9.7 and 8.9 μL, respectively after 24, 48, 72 and 96 h of treatment (Table 1).
Essential oils inhibited the oviposition response of the insect when fumigated. In oviposition inhibitory assay, oviposition was reduced to 55.39 and 28.6%; 53.97 and 32.44% and 57.81 and 28.13% of the control after treatment with 40 and 80% of 24 h LC50 of the T. ammi and A. graveolens; A. graveolens and N. sativa and N. sativa and T. ammi combination of essential oils (Table 2).
| Table 1: | Summary of Trachyspermum ammi, Anethum graveolens and Nigella sativa essential oils toxicity assays against Callosobruchus chinensis |
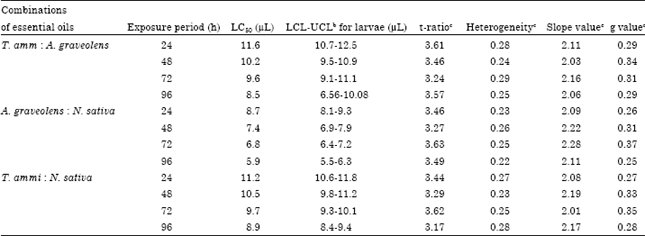 | |
| aLC50 represent the median lethal concentration. bUCL and LCL represent upper confidence limit and lower confidence limits. ct-ratio, heterogeneity, slope values and g-values are significant at all probability level | |
| Table 2: | Effect of different synergistic combinations of Trachyspermum ammi, Anethum graveolens and Nigella sativa essential oils on per cent oviposition of Callosobruchus chinensis |
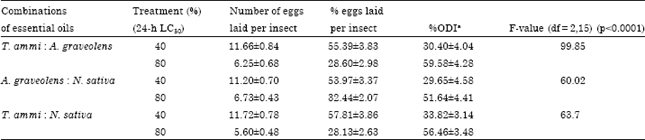 | |
| Values are represented as Mean±SD. a%ODI was calculated as 100(A-B)/(A+B), where A and B represent the number of eggs laid in control and in the test, respectively | |
The maximum per cent oviposition deterrence index (%ODI±SE) was observed in case of T. ammi and A. graveolens (59.58±4.28) followed by N. sativa and T. ammi (56.46±3.48) and A. graveolens and N. sativa (51.64±4.41). The decrease in oviposition potential of C. chinensis was significant when fumigated with the combinations of T. ammi and A. graveolens (F = 99.85; df = 2, 15; p<0.0001); A. graveolens and N. sativa (F = 60.02; df = 2,15; p<0.0001) and N. sativa and T. ammi (F = 63.7; df = 2,15; p<0.0001) essential oils (Table 2).
Chemical control using synthetic insecticides has dominated control measures against cowpea bruchids. However, these insecticides have serious problems like genetic resistance in the pest, toxic residue property of insecticides and toxicity to consumers and other non-target animals. To avoid such conditions, in recent years use of plant derived insecticides as a replacement of synthetic insecticides is gaining importance. Earlier, several successful attempts have been made by different scientific groups to use plant based insecticides to control pulse beetle bruchids (Upadhyay et al., 2006; Aboua et al., 2010; Ofuya et al., 2010). Present study is based on the fact that essential oils are complex mixture of a large number of compounds of different chemical groups and their biological effects are the results of synergism of all components. The activity of individual components of the essential oil is modulated by other components of the oil and possibly, the target site and mode of action of the essential oil components vary (Hoet et al., 2006).
Results of the present study indicate that combinations of T. ammi, A. graveolens and N. sativa essential oils have insecticidal properties against adults of C. chinensis in synergistic manner as evidenced by their low median lethal concentration values in present study as compared to efficacy of essential oils used alone in the previous study (Chaubey, 2008). In the toxicity assay, mortality rate was increased with increase in concentration of essential oils. The index of significance potency estimation, g-value indicates that the value of mean is within the limits at all probability levels (90, 95 and 99%) as it is less than 0.5. Values of t-ratio greater than 1.96 indicate that the regression is significant. The steep slope values observed in toxicity assay demonstrated that a small increase in the concentration of essential oil cause a large mortality in insects. Values of heterogeneity factor less than 1.0 denotes that model fits the data adequate.
T. ammi, A. graveolens and N. sativa essential oil combinations reduced oviposition potency in insects when fumigated with sub-lethal concentration. Ngamo et al. (2007) have reported similar results against Sitophilus oryzae when applied essential oils alone or in balanced combinations. The rapid action of essential oils against insects is indicative of their neurotoxic mode of action interfering with neuromodulator octopamine (Kostyukovsky et al., 2002) or with GABA-gated chloride channels (Priestley et al., 2003). Combinations of essential oils probably target both pathway of toxicity in insects and essential oils could be the source of active components showing synergism. Furthermore, exploration of mode of actions of essential oil constituents in combination will definitely help in understanding the mechanism of action in synergism. On the basis of observations, it can be advised for the use of T. ammi, A. graveolens and N. sativa essential oils in different combinations as insecticides as it is safe for environment, health of the consumers and economically cost effective.