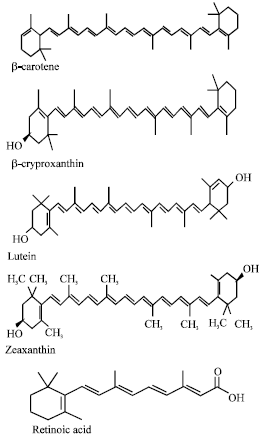
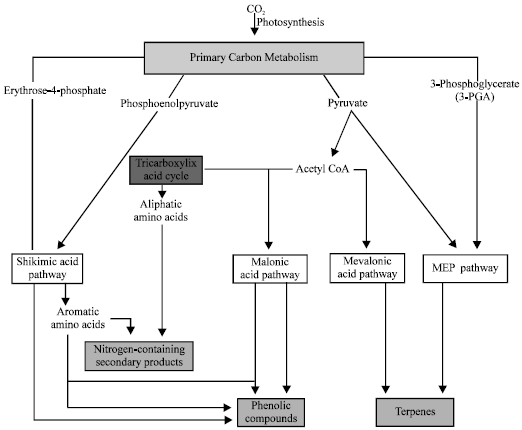
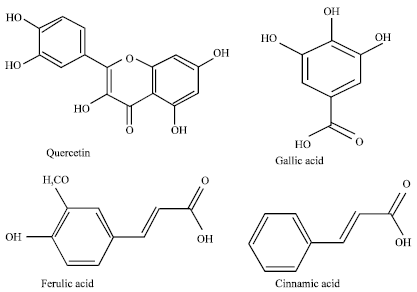
Review Article
Maize Landraces (Zea mays L.): A New Prospective Source for Secondary Metabolite Production
Plant Science Center, Plant Morphogenesis and Biochemistry Laboratory, Federal University of Santa Catarina 88040-900, P.O. BOX 476, SC, Florianopolis, Brazil
Ricardo Brasil Severino
Plant Science Center, Plant Morphogenesis and Biochemistry Laboratory, Federal University of Santa Catarina 88040-900, P.O. BOX 476, SC, Florianopolis, Brazil
Marcelo Maraschin
Plant Science Center, Plant Morphogenesis and Biochemistry Laboratory, Federal University of Santa Catarina 88040-900, P.O. BOX 476, SC, Florianopolis, Brazil