Research Article
Application of Rhizobium sp. BHURC01 and Plant Growth Promoting Rhizobactria on Nodulation, Plant Biomass and Yields of Chickpea (Cicer arietinum L.)
Department of Botany, MMV, Banaras Hindu University, Varanasi-221005, U.P., India
J. Yadav
Department of Soil Science and Agricultural Chemistry, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi-221005, U.P., India
Kavindra Nath Tiwari
Department of Botany, MMV, Banaras Hindu University, Varanasi-221005, U.P., India










Shobnath Yadav Reply
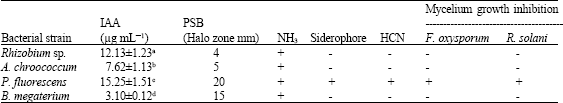
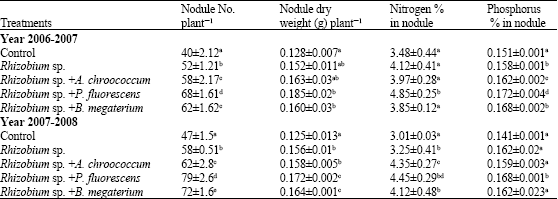
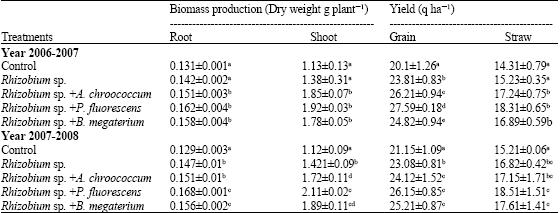
this paper work and finding are very good and appropriate combination of PGPR for chickpea production in all over world. Authors have been reported very systematic and two year field axperimentaldata. this research have directly applied research in agriculture field.
Shobnath Yadav
Research Scholar in department
of soil science and agricultural chemistry
IARI, NEW DELHI, INDIA.
Vimal Singh Reply
this paper has been reported endigenous root nodules bacteria such Rhizobium which is good symbiotic relation with chickpea crops. this paper provide good information about PGPR with rhizobium relation and synergistic combination for all chickpea crops for high yield.
Vimal Singh
research scholar
Department of Biotchnology
Faculty of Science
BHU, Varanasi-221005
Jay Prakash Verma
thank you for your positive response and good information about my research.
thanks