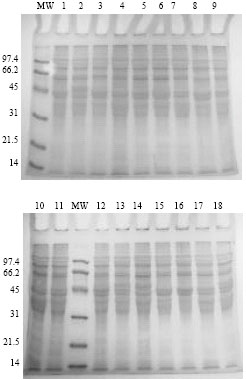
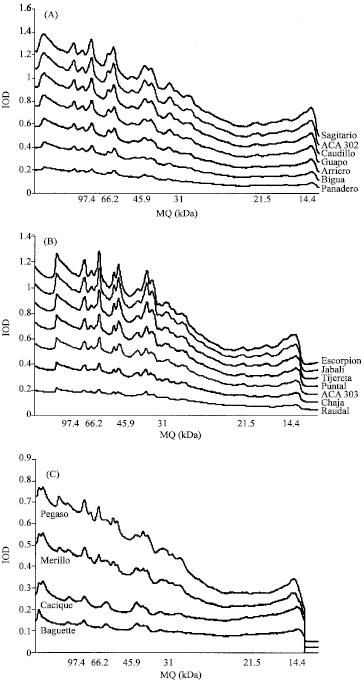
Research Article
Relationship Between Variety Classification and Breadmaking Quality in Argentine Wheats
CONICET, Argentina
Gabriela T. Perez
CONICET, Argentina
Pablo D. Ribotta
Facultad de Ciencias Agropecuarias, Universidad Nacional de Cordoba, CC 509, 5000 Cordoba, Argentina
Alberto E. Leon
Facultad de Ciencias Agropecuarias, Universidad Nacional de Cordoba, CC 509, 5000 Cordoba, Argentina