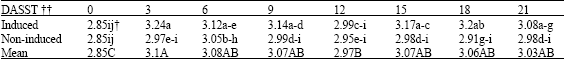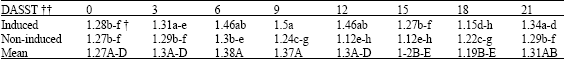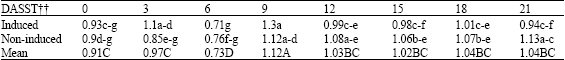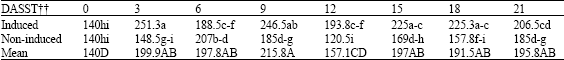Research Article
Changes in Mineral Nutrition Levels during Floral Transition in Strawberry (Fragaria x ananassa Duch.)
Department of Horticultural Science, College of Agriculture, Shiraz University, Shiraz, Iran
E. Tafazoli
Department of Horticultural Science, College of Agriculture, Shiraz University, Shiraz, Iran