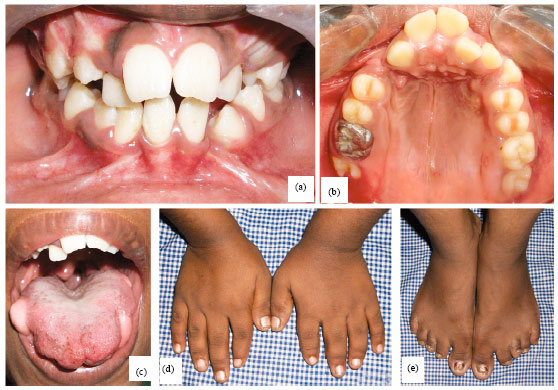
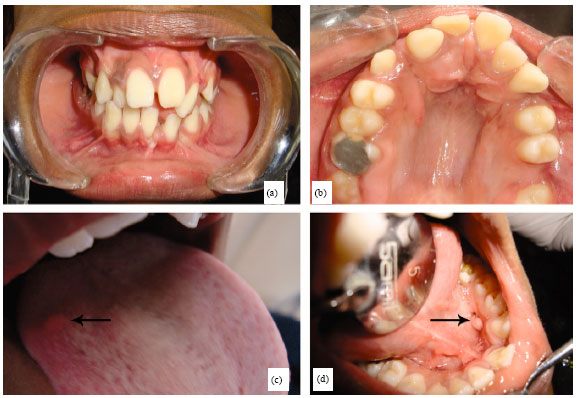
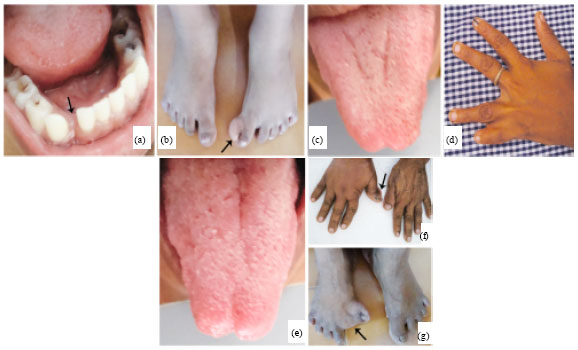
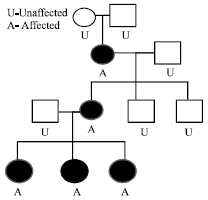
Case Study
Hereditary Oro-facial Digital Syndrome Type 1: Diagnosis and Management-case Report
Department of Pediatric Dentistry, Amrita School of Dentistry, Cochin, India
Kavita Rai
Department of Pedodontics and Preventive Children Dentistry, A.B. Shetty Memorial Institute of Dental Sciences, Deralakatte, Mangalore 18, India
Amitha M. Hegde
Department of Pedodontics and Preventive Children Dentistry, A.B. Shetty Memorial Institute of Dental Sciences, Deralakatte, Mangalore 18, India
Saurabh Joshi
Department of Pedodontics, Rural Dental College, Loni, Maharashtra, India