Research Article
The White Cliffs of Dover are an Example of Natural Carbon Sequestration
Department of Biology, Monmouth University, 400 Cedar Ave, West Long Branch, NJ 07764, USA
During a scientific meeting at Monmouth University in April 2010 the subject of Global Warming (Ackerman and Knox, 2006) and its attenuation by carbon sequestration was considered. In particular the role of algae was discussed because it is strange but true that small creatures can have very large effects on ecosystems. One group is the coccolithophores (Green and Leadbeater, 1994) which are beyond clear observation by light microscopy but whose effects are very large because of their cell wall chemistry (Mukkamala et al., 2006). Coccolithophores are unicellular algae, common in oceanic ecosystems (Reid et al., 2009), which have a distinctive feature: cell walls composed of calcium carbonate plates (coccoliths, Fig. 1). Late in growth, blooms of these algae shed large amounts of their coccoliths. These turn water a milky turquoise color (Shutler et al., 2010) similar to the rock flour of glacial lakes (personal observation). This material is easily detected in satellite images because of its ability to backscatter visible light (Merico et al., 2003; Smyth et al., 2004) against a background of more transparent sea water. Their process of cell wall formation involves catalyzing the simple reaction of carbon dioxide (carbonic acid) to carbonate. This is a spontaneous reaction familiar to any scientist that has prepared a solution of sodium hydroxide then stored the solution in a bottle with a loose cap; a fine white precipitate of sodium carbonate is formed by atmospheric CO2 reacting with the NaOH.
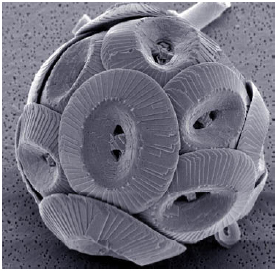 | |
| Fig. 1: | Coccolithus pelagicus (Lampitt, 2008), showing button shaped cell wall plates composed of calcium carbonate. This species is common in cool North Atlantic waters (Cachao and Moita, 2000) |
As with most life processes coccolith formation is an enzymic process and coccolithophores do not cause the reaction, but they do greatly increase the rate of reaction.
Much was learned about coccolith sedimentation and metamorphosis to calcite during the boom in North Sea Oil exploration of the 1960's and 1970's. Coccoliths are not significantly larger than the fine carbonate powders produced in chemical reactions and sediment at about 13.8 cm day-1 (Steinmetz, 1994), so slowly that they may drift to unsaturated zones of the ocean which allow carbonate dissolution (Honjo, 1975) before fine carbonates can settle to the sea floor. However, because coccolithophore algae are consumed by animals such as copepods and aggregated into fecal pellets (Ziveria et al., 2007) they become sufficiently large to sediment rapidly (200 m day-1, Steinmetz, 1994), accumulate and metamorphose to the mineral chalk, a form of cemented calcite particles (Neugebauer, 1974). This displaces the sea surface chemical equilibrium, drawing more CO2 into reaction to carbonate. Rapid coccolith aggregate sedimentation is a process that has sequestered large amounts of CO2 in the geological past (Gregory, 2002) and is a factor for enormous and rapid reduction of atmospheric CO2 and therefore global warming (Fig. 2, Salter et al., 2007). Even so, coccoliths may suffer partial or complete dissolution after reaching ocean sediments (Roth and Berger, 1975) because deep sea water may not be saturated with carbonate.
These microscopic biological activities might be ignored by most people if it were not for their large scale effects that are visible to the unaided eye: for example, the White Cliffs of Dover (Fig. 2, Table 1). The cliffs are composed of fossilized coccolith aggregates (Saruwatari et al., 2008) that fell to the bottom of shallow seas and built up deposits which were mineralized to the chalky form of calcium carbonate (Mukkamala et al., 2006). This was an important natural process in warm shallow seas (Buitenhuis, 2000), when thick layers of calcite formed on European continental shelves when their margins were flooded (Katz et al., 2004; Kennedy and Garrison, 1975) during the cretaceous epoch (Tyson and Funnell, 1987). The White Cliffs are prominent, but they are just the tip of an iceburg of calcite deposits that extend over large areas of North Western Europe (Fig. 3) (Hakansson et al. (1991), Buitenhuis (2000) and citations within); naturally sequestered carbon, removed from the carbon dioxide rich atmosphere of the cretaceous epoch.
 | |
| Fig. 2: | The White Cliffs of Dover near the South Foreland Lighthouse, seen from St. Margaret's Bay, Dover, 51°8'6" N, 1°22'30" E (Jouan, 2006). The Lighthouse is 21 m high and the cliffs are 101 m high at that point (Clegg, personal communication) |
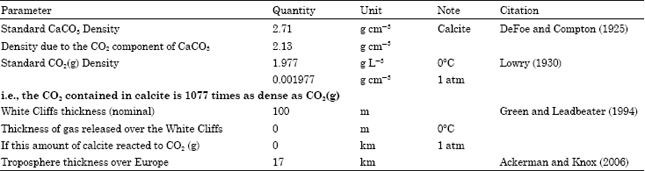
| Table 1: | Physical features of carbon dioxide sequestered as calcite (De Foe and Compton 1925, Lowry 1930) |
 | |
GLOBAL ENGINEERING AND UNINTENDED CONSEQUENCES
Coccolithophores have long been recognized as unique and interesting algae (Siesser, 1994), but recently they have attracted interest from global engineering theorists. Ecologists with ambitions for global environmental engineering have looked at the White Cliffs of Dover and been inspired by the effects of coccolithophores (Woodward et al., 2009). Their ambition is to achieve similar large scale Carbon Sequestration and control atmosphere composition (Wilson and Gerard, 2007; De Baar et al., 2005). These scientists see the contribution by fossil fuels of 5.4x1012 tonnes of carbon per year to the atmosphere as a problem that could be solved if the converse process by coccolithophores was induced i.e., consignment of 3.2x1012 tonnes carbon per year as carbonate to sediments (Raven and Falkowski, 1999). These are comparable figures and might be used like tunable factors to balance one another and control atmospheric CO2 content. The most commonly suggested method for achieving this process is stimulating blooms of coccolithophores by ocean fertilization with inorganic iron (De Baar et al., 2008; Jin et al., 2008) because iron is the limiting nutrient in oceanic ecosystems (Pomar and Hallock, 2008) i.e., creating Artificial White Cliffs of Dover by stimulating coccolithophore blooms.
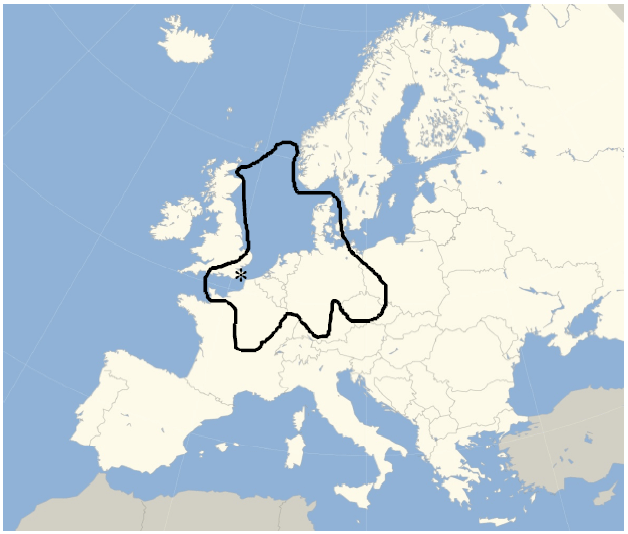 | |
| Fig. 3: | Western Europe, showing the approximate extent of calcite deposits (Downing et al., 1993) that formed as continental shelves were inundated (Tyson and Funnell, 1987; Hakansson et al., 1991). The White Cliffs of Dover are indicated by "*". These deposits formed during the cretaceous epoch, a name derived from the Latin for chalk; creta |
However several nutritional and other factors determine coccolithophore blooming (Tyrrell and Merico, 2004) and other algae may also form blooms near the coccolith growth optima (Margalef, 1978; Balch, 2004), making the control of coccolithophore bloom processes tenuous. Observing field work is also limited by detecting coccolithophore bloom as scattered light: it is not only coccoliths that create significant back scatter (Tyrrell and Merico, 2004). Diatoms are significantly stimulated by iron fertilization (De Baar et al., 2005) and have highly refractive silica cell walls. Therefore measurements that rely on satellites only must be treated with caution.
While the sight of the White Cliffs is inspiring it may be salutary to remember how common the unintended consequences of our actions are (Hu et al., 2003). Algal blooms, including blooms of coccolithophores are common, but how algae thrive or die and how they bloom is a generally unknown process and not easily manipulated by us in a controlled way (Marsh, 2003). Many of the methods of geochemical engineering have strong critics and increasing numbers of objections have been made (Robock, 2008; Robock et al., 2009; Hegerl and Solomon, 2009). This may be because of the very complex nature of both atmospheric and biological systems. For example, Raven and Falkowski (1999) list twenty two processes or factors involved in the movement of carbon from the atmosphere to oceans and sediments i.e., these are difficult systems to understand even if all relevant factors are independent and linear (simple to extrapolate), or even known. Some particular unintended consequences of coccolithophore blooms are already known (Denman, 2008; Malin and Steinke, 2004) and include increased but unquantified denitrification and production of the long lived greenhouse gas N2O (Law, 2008) i.e., plans to reduce carbon dioxide using coccolithophores would result in increases in other greenhouse gases. This is a paradox that has been seen before, for example carbon sequestration by increased forestation may, in some ecosystems, result in increased methane emission (Megonigal and Guenther, 2008), another greenhouse gas.
NATURAL PROCESSES ARE IN STABLE EQUILIBRIA
Part of the problem of global environmental engineering is an assumption that the climate is characterized by unstable equilibria which are capable of runaway changes to new stable states. However, if this were true the earth's climate could not recover from the many great knocks it has experienced in the past. Examples include volcanic eruptions which have covered millions of square kilometers in molten basalt (Pande, 2002) or darkened the atmosphere with large volumes of particulate matter (Schroder, 2002), which are later washed out by rain (Park et al., 2005). When the earth has been violently struck by comets there was an initial perturbation (Kring, 2007) then a resumption of our present desirable climate equilibrium, not a runaway and permanent disaster. In fact, asteroid impacts may be a regular occurrence in the history of the earth due to the passage of the solar system through galactic debris belts and life has always recovered (Bailer-Jones, 2009). These violent events indicate a stable rather than unstable equilibrium between factors controlling the climate; like a pendulum displaced from equilibrium, the atmosphere naturally swings back from extremes. The atmosphere is not like a tall vase which may teeter when knocked, then fall and occupy a second horizontal state of equilibrium, far from the first. If there's many a slip twixt cup and lip it is better to rely on natural processes, such as those that formed the White Cliffs of Dover and the stable equilibrium of geological processes we have all benefitted from for so long, than the uncertain outcomes of global engineering plans which have not been adequately formulated or tested.
John Tiedemann and Dennis Rhoads inspired this study. Fang Xie of the Guggenheim library at Monmouth University was very helpful in preparation of this study.