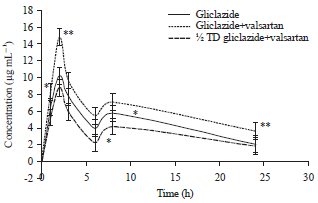
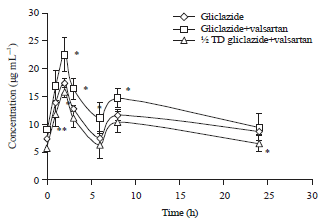
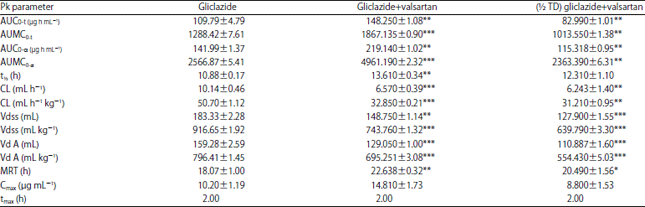
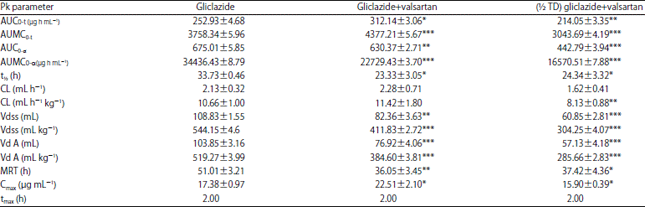
Research Article
Effect of Valsartan on Pharmacokinetics and Pharmacodynamics of Gliclazide in Diabetic rats
Department of Pharmacology, Jyothishmathi Institute of Pharmaceutical Sciences, Beside LMD Police Station, Thimmapur, 505481 Karimnagar, Telangana State, India
LiveDNA: 91.1516
B. Vijay Kumar
Department of Pharmacology, Jyothishmathi Institute of Pharmaceutical Sciences, Beside LMD Police Station, Thimmapur, 505481 Karimnagar, Telangana State, India