Research Article
Optical Density Utilization in Sunflower (Helianthus annuus L.) Seed Extracted Protein Evaluation and Characterization
Esfahan Payamenoor University, Esfahan, Iran
Fazilati Mohammad
Esfahan Payamenoor University, Esfahan, Iran
The sunflower is one of the four most important oil crops globally and is grown on over 25 million hectares worldwide. Seeds of sunflower are mainly used for their oil content which accounts for 80% of the value of the sunflower crop. At the same time, there is an increasing interest in the use of sunflower protein (Helianthus annuus L.) in human nutrition. Sunflower seeds contain up to 20% of protein, whereas protein contents of the oil press cakes and extraction residues range from 30-50% (Dorrell and Vick, 1997). The techno-functional properties of sunflower proteins are comparable with those of soy and other leguminous proteins (Gonzalez-Perez et al., 2005). Although lysine deficiency is a major drawback from the nutritional point of view, proteins from sunflower press cake are considered a valuable alternative as food ingredients, since they are low in antinutritional compounds and devoid of toxic substances (Gonzalez-Perez and Vereijken, 2007). The sunflower protein isolates can be subjected to hydrolytic treatments in order to produce protein hydrolysates that have improved functional and nutritional properties (Villanueva et al., 1999). In addition, protein hydrolysates are also a source of bioactive peptides which are short peptides and have a certain biological activity that may be beneficial for the organism (Megias et al., 2008). Bioactive peptides with different impacts on the regulation of the gastrointestinal,nervous, cardiovascular and immune systems have been described (Korhonen and Pihlanto, 2006). Composition and conformation are responsible for a sunflower protein’s functionality. Compositional differences that may alter functionality include the ratio of protein fractions, variations in subunit concentrations within fractions and differences in amino acid profiles. Sunflower proteins have two major salt-extractable fractions (2S and 11S) that can be isolated on the basis of their sedimentation coefficients. The main storage proteins in sunflower make up about 85% of the total protein content. The 11S helianthinin belongs to the widely distributed legumin-like family of globulins and 2S albumins which, in turn, belong to a larger protein family. The helianthinins are generally considered to be the major group of storage proteins,being reported to account for some 60% of the total proteins in the mature seed with the 2S albumins accounting for about 20% (Kortt and Caldwell, 1990). However, early studies of the salt-soluble proteins by sucrose gradient centrifugation gave different results, with 62% 2S albumins and 38% 11S globulins, Youle and Huang (1981) and Gonzalez-Perez and Vereijken (2007) reported that globulins constitute most of the sunflower proteins, ranging from about 40-90% while albumins account for about 10-30% of the total proteins. Glutelins and, in particular, prolamins are only minor fractions. The proportions of proteins with different sedimentation coefficients depend largely on conditions such as type of buffer, pH, ionic strength, chemical, physical or enzymatic modification, etc. (Molina et al., 2001). Therefore, literature data show considerable variation in the proportions of the different protein fractions according to the sedimentation coefficient. Also, sunflower proteins have two small amount salt-extractable fractions (6-9S) and high molecular weight fraction (15-18S). The latter fraction has been described as an aggregate of the 11S fraction (Gueguen et al., 1988). In contrast to soybean globulins sunflower globulins do not contain any genetically independent 7S constituent (Anisimova and Gavrilyuk, 1990). Nevertheless, various amounts of proteins with 7s sedimentation coefficient have been detected in sunflower seeds (Kabirullah and Wills, 1983). These 7S constituents are most likely dissociation products of the 11S globulins, a dissociation which also occurs in soybean glycinin. In addition to the storage albumins and globulins, sunflower seeds also accumulate a third group of proteins. These are the oleosins, or oil body proteins which are associated with the outer surface of the oil bodies and are thought to prevent coalescence (Ross and Murphy, 1992). Helianthinin has been reported to be present as a globular oligomeric protein with a Molecular Weight (Mw) of 300-350 kDa. The currently most accepted model of helianthinin (11S), at neutral pH, consists of an arrangement of six spherical subunits into a trigonal antiprism (Plietz et al., 1983). The monomeric subunits consist of an acidic (32-44 kDA) and a basic (21-27 kDa) polypeptide linked by a disulfide bond (Dalgalarrondo et al., 1984). Sunflower albumins are basic proteins with a molecular mass in the 10-18 kDa range (Kortt and Caldwell, 1990). Two types of albumin are distinguished: Methionine-rich and methionine-poor albumins. In contrast to 2S seed albumins from other species that consist of two chains linked by disulfide bonds, sunflower albumin consist of a single polypeptide chain (Pandya et al., 2000). Sunflower protein contains low levels of lysine, whereas it is relatively rich in sulphur-containing amino acids compared with other oil seeds (Canibe et al., 1999). These authors reported that the average contents of lysine, threonine, cystine and methionine in twelve sunflower genotypes were 3.72, 3.65, 1.63 and 2.33 g/16 g N, respectively. Glutamic acid was the amino acid present at the highest concentration (19.18 g/16 g N). The purpose of the present study was to employ analytical methods to determine differences among investigated sunflower genotypes and whether the analyzed hybrids could be sources of specific proteins. A more detailed knowledge of the variability of protein and protein subunit accumulation among sunflower genotypes could facilitate ongoing efforts to improve both quantity and quality of sunflower protein. Seed storage proteins may be used to determine the cultivar trueness and genetic purity of the sample.
Plant material: The six oilseed hybrids Lotto3620, Lotto7R09, LottoR101, Lotto7R0, LottoR28, mt8, TR3 and Cms8 of sunflower (Helianthus annuus L.) selected for this investigation were obtained from National Institute of Genetic Engineering and Biotechnology (NIGEB),Tehran, Iran. All plant materials are adapted to conditions and have been developed in Iran. Seeds were collected at full maturity from plants grown in a field trial at the during the growing season. The defatted whole meal flour (particle size <500 μm), obtained by grounding sunflower seeds. Defatting was carried out by diethyl ether extraction at 35° in a Soxhlet extractor. Albumin, globulin, prolamin and glutelin contents Different protein fractions were obtained by successive extractions of defatted sunflower flour with a series of solvents (in a ratio 1:10 w/v) according to a modified Landry and Moureaux (1970) method. Distilled water, 0.5 M NaCl, 70% ethanol and 0.2 M NaOH were used to extract albumin, globulin, prolamin and glutelin fractions, respectively. Extraction of each protein fraction was done by repeated stirring tree times for 30 min at 4°C, followed by centrifugation at 20,000 g for 15 min. Protein content was calculated in each fraction from the nitrogen content determined by the micro Kjeldahl method using 5.50 as the conversion factor. The results are given as percentage of Dry Matter (d.m.) as well as percentage of total protein (protein solubility index-NSI. (Sodium dodecyl sulfate-polyacrilamide gel electrophoresis Soluble protein composition of the defatted samples was detected by the Sodium Dodecyl Sulfate-polyacrilamide Gel Electrophoresis (SDS-PAGE) performed according to Fling and Gregerson (1986) on 12.5% gel in vertical electrophoretic unit (LKB, Sweden). Defatted flour was extracted for 120 min at room temperature with Tris-HCl buffer pH 8.0 in the ratio 1:20 and centrifuged at 17,000 g for 15 min (Thanh and Shibasaki, 1976). The protein content in the supernatant was determined according to the method of Bradford (1976) using bovine serum protein (BSA, Sigma, USA) as a standard (Table 1).
| Table 1: | Bradford assay and Bovin Serum Albomin (BSA) concentration |
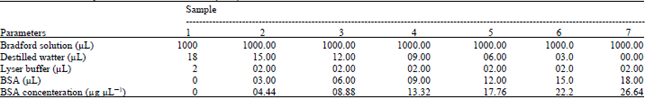 | |
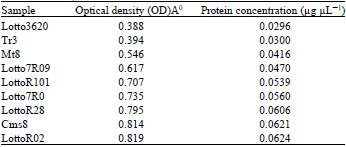
By means of Bradford assay hybrids extracted protein concentration was evaluated according to the equation: y = 76.29x+0.001 (Fig. 1). Optical density for Lotto3620, TR3, mt8, Lotto7R09, LottoR101, Lotto7R0, LottoR28, Cms8 and LottoR02 was 0.388A0, 0.394A0, 0546A0, 0.617A0, 0.707A0, 0.735A0, 0.795A0, 0.814A0 and 0.819A0, respectively. The lowest Optical Density (OD) observed in Lotto3620 and highest optical density observed in LottoR0 (Table 2 and Fig. 3).
Prior to electrophoresis, soluble proteins have been diluted to 2 mg cm-3 with the sample buffer (0.055 M Tris-HCl, pH 6.8, 2% (w/v) Sodium Dodecyl Sulfate (SDS), 7% (v/v) glycerol, 4.3% (v/v) β-mercaptoethanol, 0.0025%), w/v) bromophenol blue), heated at 90°C for 5 min and cooled at room temperature. Twenty five microliter sample was loaded per well. Gels were run at 30 mA for 6 h, fixed and stained with 0.23% (w/v) Coomassie Blue R-250 dissolved in 3.9% (w/v) Trichloroacetic Acid (TCA), 6% (v/v) acetic acid and 17% (v/v) methanol for 45 min. Destaining was performed with 8% acetic acid and 18% (v/v) ethanol. Molecular weights of the polypeptides were estimated by using low molecular weight standards ovatransfrin (77.000 kDa), ovalbumin (45.000 kDa), carbonic anhydrase (30.000 kDa) and myoglobin (17.200 kDa). The protein bands on the destined gel were quantitated using SigmaGel software version 1.1 (Jandal, San Rafael, CA) (Fig. 2).
The optical density ratios according to the lowest Optical absorption to which belonged Lotto 3620, was 1, 1.0154, 1.407, 1.590, 1.822, 2.048, 2.097, 2.11, in the sunflower hybrids, Lotto3620, Tr3, Mt8, Lotto7R09, LottoR101, Lotto7R0, LottoR28, Cms8 and LottoR02, respectively (Fig. 4). However, the content of protein fractions, subunits and different polypeptides are expressed as percentage of total extracted protein. Therefore, our data are a result of differences in optical density absorption among hybrids.
| Table 2: | Optical density and protein concentration for six hybrids and Tr3, Cms8, Mt8 |
 | |
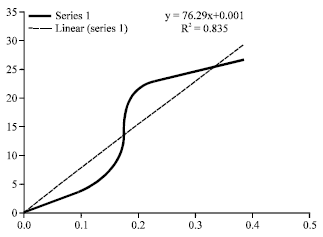 | |
| Fig. 1: | Optical density regression curve according to seven sample BSA concentration |
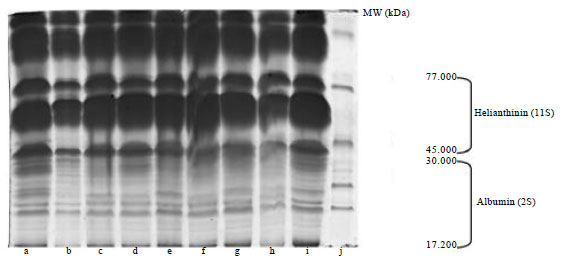 | |
| Fig. 2: | SDS-PAGE patterns of proteins related to (a) Lotto7R0, (b) Tr3, (c) Mt8, (d) Cms8, (e) Lotto7R09, (f) LottoR02, (g) Lotto3620, (h) LottoR101, (i) LottoR28 and (j) Ladder |
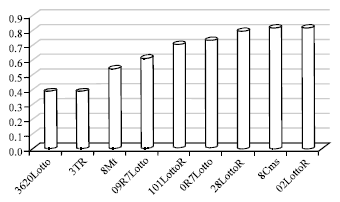 | |
| Fig. 3: | Optical density (A0) in six sunflower hybrids and Cms8, Mt8, Tr3 |
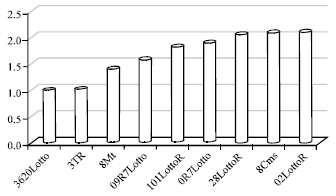 | |
| Fig. 4: | Optical density ratios in six sunflower hybrids and Cms8,Mt8, Tr3 according to the lowest absorption in Lotto3620 with 0.388A0 |
Sunflower protein applications refer mainly to the fortification of foods by sunflower meal, especially meat and milk products, infant formulae, bakery products and pasta products. Data in Table 2 indicated that there is an ascending process in protein concentration among six hybrids, Cms8, Mt8 and Tr3 also. The results indicated that the most protein concentration observed in LottoR02 and consequently the highest optical density belonged to LottoR02 with 0.819A° while the minimum protein concentration observed in Lotto 3620 and consequently the lowest optical density belonged to Lotto3620 with 0.388A°. Water soluble proteins, determined by the Landry and Moureaux (1970), were the dominant protein fraction in all the genotypes. Considering that most water soluble proteins are also soluble in salt solution, the literature notes the helianthinin fraction as the major storage protein component in sunflower that accounts for approximately 40-90% of the total storage proteins in sunflower seed (Gonzalez-Perez and Vereijken, 2007).
Electrophoresis patterns of total proteins from the analyzed genotypes are shown in Fig. 2. The concentration of total extracted proteins, determined by Bradford (1976) method, was 0.029, 0.0300, 0.0416, 0.0470, 0.0539, 0.0560, 0.606, 0.0621 and 0.0624 μgr μL-1 in the seeds of the Lotto3620, Tr3, Mt8, Lotto7R09, Lottor101, Lotto7R0, LottoR28, Cms8, LottoR02, respectively. However, 20 μL of the solution were loaded per well. The quantification of the 11S and 2S fractions and the other detected polypeptides was done by densitometric analysis of the gel protein fraction. The SDS-PAGE profiles of the seed proteins in the crude extracts for all the genotypes showed a very similar number of protein bands in the electrophoretograms (Fig. 2). The highest molecular weight protein observed in the seed was 77 kDa and the lowest 17.200 kDa. Jiang et al. (1994) reported that sunflower proteins showed bands at 21-57 kDa. Figure 2: SDS-PAGE patterns of proteins from sunflower hybrids from 17.200 to 30.000 kDa and 30.000 kDa to 45.000 indicate basic and acidic polypeptides of 11S helianthinin, respectively. A treatment with the reduction agent β-mercaptoethanol induced dissociation of the helianthinin fraction to polypeptides whose spectrum included three groups of components, two of which were acidic (α, Mw = 36.8-42.9 kDa and α', Mw = 31-35 kDa) and one basic (β, Mw = 21-29.6 kDa) (Fig. 2). The results of Anisimova et al. (2004) showed that the standard helianthinin spectrum included eight to ten major components and a number of minor variants. Association and dissociation phenomena are a common feature of many 11S seed globulins (Marcone, 1999). Like other 11S seed globulins, helianthinin seems to dissociate into subunits according to the following scheme: 11S→7S→3-2S (Gonzalez-Perez et al., 2004). The molecular weight of the 2S albumin proteins ranged from 11.5-20.1 kDa. According to Kortt et al. (1991) the sunflower 2S protein fraction contains at least eight distinct proteins and two of these are sulfur-rich with 8% of residues cysteine and 16% methionine. In our study, the highest optical absorption observed in LottoR02 with 0.819A0 and lowest optical absorption observed in Lotto3620 with 0.388A0 (Fig. 3). In all analyzed samples, the optical absorption ratios were 1,1.0154, 1.407,1.590, 1.822, 1.894, 2.048, 2.097, 2.110 for sunflower Lotto3620, Tr3, Mt8, Lotto7R09, Lottor101, Lotto7R0, LottoR28, Cms8 and LottoR02, respectively (Fig. 4). These results indicated that the highest protein concentration belonged to LottoR02 and lowest protein concentration belonged to Lotto3620. The ratio of 11S/2S proteins varied from 2.08 t according to Mazhar et al. (1998). The mean 11S helianthinin to 2S albumin ratio was 2:1. This ratio is known to influence the protein quality of sunflower and greatly affects the functional properties of food products made from sunflower (Gonzalez-Perez et al., 2005; Kortt et al., 1991) reported that the sunflower 2S proteins are resistant to degradation by rumen bacteria. This indicates that these sulfur-rich proteins may be ideal candidates for improving the nutritive content (with respect to the sulfur amino acids) of the seed and vegetative tissue of plants destined for ruminant feeding. However, sunflower seed has been responsible for serious anaphylactic reactions in some allergic individuals. The nature of the major allergen remains unknown but some IgE-binding proteins have been identified, including the 2S methionine-rich albumin protein (SSA). Preliminary findings indicated the possibility that SSA possesses linear epitopes (Kelly and Hefle, 2000). The globulins (helianthinin) have typically been obtained as a secondary by-product from the processing of the seed.The protein isolates in human foods continue to be used as a meat substitute in products such as hamburgers and sausages and have also been used as whipping agents, emulsifiers and binding agents in a variety of food products such as bakery products and dairy analogues in order to replace more expensive animal-based protein ingredients derived from eggs and milk (Fukushima, 1991). There is not much data in the literature on the content of sunflower protein fractions, so it was difficult to compare our results. Different problems arose during the isolation and purification step of the major protein fraction of sunflower seeds (Durante et al., 1989). One of the reasons was the presence of relatively high amounts of phenolic compounds, especially chlorogenic acid. Phenolic compounds interact and form complexes with proteins, thereby reducing both their digestibility and their functionality (Sastry and Rao, 1990). Many methods have been proposed for isolating sunflower protein and removing phenolic compounds from sunflower seeds. Also, one of the main factors that has complicated the purification and characterization of globulins (helianthinin) is that they are known to dissociate with very small shifts in pH (Wright, 1987). It therefore appears that pH dependent dissociation of globulins is a common physicochemical characteristic. Our data provide increased knowledge of the sunflower proteins.
Currently, limited information is available on the biochemical and genetic mechanisms that regulate high-proteins. Essentially, two avenues of improving sunflower protein have to be utilized. The first is through traditional breeding using high-protein germplasm and the second is the use of biotechnology. The results showed that the protein bands were similar among all the sunflower samples. Three polypeptides groups of helianthinin fraction were detected. Two of these were acidic (α, Mw = 36.8-42.9 kDa and α', Mw = 31-35.3 kDa) while one was basic (β, Mw = 21-29.6 kDa). The molecular weight of the 2S albumin proteins ranged from 11.5 to 20.1 kDa. However, the contents of 2S and 11S proteins were statistically different among the sunflower hybrids. Our data provide increased knowledge of the variability of protein and protein subunit accumulation among hybrids which will facilitate ongoing efforts to improve both the quantity and quality of sunflower protein.