Research Article
In vitro Screening of Chewing Stick Extracts and Sap on Oral Pathogens: Immune Compromised Infections
College of Medicine, Nigeria
O.A. Odukoya
University of Lagor, Nigeria
A.F. Otegbeye
Idi Araba, Lagor, Nigeria
Chewing sticks have been used for oral hygiene by many cultures since antiquity. This is particularly prevalent in Nepal, India, Africa and Malaysia, especially in rural areas and among the people of low socio-economic class because they are cheap and easily available (Almas, 2001).
Several studies have shown that these chewing sticks in addition to the mechanical cleansing action also possess certain substances like fluoride, silicon, alkaloid, essential oils, tannins, resins, gum and anthraquinones. These substances reduce bacterial colonization and plaque formation thereby inhibiting caries formation, stimulate oral tissues and control bacterial growth in the oral cavity (Fadulu, 1975; Akpata and Akinrimisi, 1997; Odebiyi and Sofowora, 1979; Wolinsky and Sote, 1984). Plants used as chewing sticks grow wild in Nigeria. The stem or root of the plant is commonly used as chewing sticks. The immunocompromised state, whether it is the result of chemotherapy, immunosuppressive therapy, AIDS, or radiation therapy has certain features in common. All immunocompromised patients are susceptible to oral infections, intraoral bleeding and may have unfavourable change in their oral mucosa. As a consequence of these complications, management of all immunocompromised patients follows a similar pattern. The commonest lesions are candidiasis and herpes viral infections, but others include ulcers, periodontal disease and malignant neoplasms. Purpura and spontaneous gingival bleeding also are seen in patients with leukaemia (Davies, et al., 2000). Thrush and erythematous candidiasis are common in patients with immune defects and are often an early manifestation of the immunodeficiency. There is an increase, especially in those with HIV infection or AIDS, in antifungal resistance of Candida albicans and in non-albicans species such as C. krusei and new species such as C. dubliniensis and C. inconspicua. Immunocompromising conditions predispose patients to oral leucoplakia and carcinoma Kaposi’s sarcoma and lymphomas. The yeast C. albicans causes oral thrush, vaginitis andinfection of skin and nails. In immune compromised patients it 1.04 causes pulmonary and generalized infections, which can be fatal. HIV patients are also prone to developing severe oral ulcers that often get infected leading to a high morbidity.
In situations where antifungal drugs and antibacterial mouthwashes cannot be procured either due to unavailability, high cost or low purchasing power of the patient (especially HIV/AIDS patients and people in rural areas) the use of these chewing sticks will offer enormous potential in managing these diseases. Dentists are now more frequently asked to provide dental care for immunocompromised patients. The traditional hospital oral care protocol includes the use of chlorhexidine, hydrogen peroxide, sodium bicarbonate, thymol glycol, benzocaine mouthrinse and nystatin.
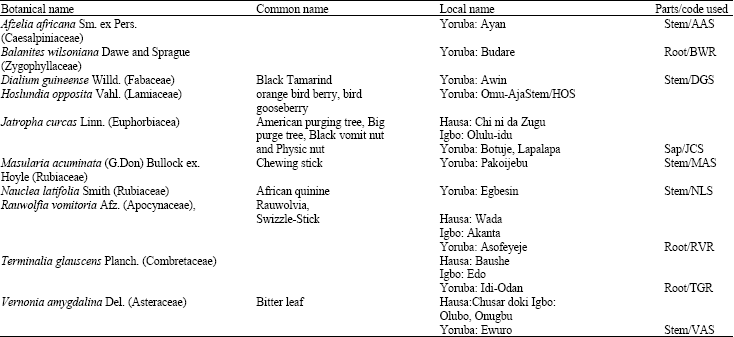
This study aims to investigate the antimicrobial and antifungal properties of J. curcas sap and aqueous extracts of nine selected indigenous chewing sticks A. africana, B. ilsoniana, D. guineense, H. opposita, N. latifolia, M. acuminata, R. vomitoria, T. glauscens and V. amygdalina on some bacteria and fungi isolated from the oral cavity with a view of exploiting the extracts as affordable sources of antifungal/antibacterial mouthwash/toothpaste.
Collection of plant materials and preparation of extracts: The various shrub/stem/roots used as chewing sticks were purchased as chewing sticks samples tied into small bundles from sellers in an open market, Lagos, Nigeria. J. curcas sap was collected from a J. curcas tree in the premises of the College of Medicine, University of Lagos. The Forest Research Institute of Nigeria (FRIN) herbarium confirmed the species identity. The samples were cut into small pieces and dried at 50°C. Further disintegration was achieved with household wooden mortar and pestle.
Preparation of extract and sap: Ten 8 grams each of the dried and powdered material was soaked in 100 mL of portable water for 24 h and filtered to produce a 10% extract for the antibacterial assay; this process was repeated to produce a 50% extract. The sap of J. curcas collected was stored in the refrigerator until when needed.
Culture media: Nutrient agar (oxide) was used for the bacterial medium. For candida, Saboraud’s dextrose agar, blood agar, chocolate agar and nutrient agar were used.
Sources of micro organisms: Bacterial isolates were obtained from mouth washings collected from 50 patients made up of 32 healthy and 18 sick patients at the Dental Center of the Lagos University Teaching Hospital. The patients were given distilled water to rinse their mouth and then spit it into sterile universal bottles; the samples thus collected were then analyzed in the laboratory.
Culture of samples: The samples were shaken thoroughly and 0.02 mL of the samples were inoculated on various plate media such as blood agar, MacConkey agar and chocolate agar using sterile standard wire loop. The plates were then incubated aerobically at 37°C for 24 h after which they were examined for growth. Those showing growth were read, taking note of their colonial characteristics and ones suspected to contain candida were sub-cultured onto a selective media- Saboraud’s dextrose agar and incubated aerobically at 37°C for 24 h, the isolated were Gram-stained to confirm it was yeast. The organisms present were finally identified by standard biochemical tests (Cowan and Steel, 1993).
Agar diffusion method: Two or three well-isolated colonies on blood agar plates were removed with sterile wire loop and transferred into a well-prepared Nutrient agar already bored with a core borer. 0.03 mL of the extract aseptically drawn was delivered into each of the hole using a Pasteur pipette calibrated to deliver 0.02 mL per drop. The drop absorbed into the agar and was incubated at 37°C aerobically for 48 h. The zones of inhibition (mm) were measured using calibrated rule.
Agar dilution method: Serial double-fold dilutions of the extracts that inhibited the growth of the organisms were prepared using the broth dilution method. The dilution was made in seven-fold giving ½, 1/4, 1/8, 1/16, respectively in test tubes and each dilution was inoculated with the test organisms and incubated overnight at 37°C. The next day the incubated dilution were sub cultured on chocolate agar plate and incubated for 48 h at 37°C and the result was recorded. Two control tubes were maintained for each test batch. These included antimicrobial control (tube containing extract and the growth medium without inoculum) and organism control (the tube containing the growth medium, physiological saline and the inoculum).
Forty Eight (96%) of the total samples colleted yielded microbial growth. Out of these, 30 (60%) are from healthy patients and 18 (36%) from sick patients. Based on the number of isolates, it was observed that 18 (100%) of the samples from the sick patients yielded growth while 30 (96%) of the samples from healthy patients yielded growth. 2 (4%) of the samples from healthy patients did not yield any growth. 21 (70%) samples from healthy patients yielded microbial pathogens and 9 (30%) yielded normal flora. From the sick patients, 16 (89%) samples yielded mainly pathogens while 2 (11%) yielded Brahamella catarrhalis and Streptococcus viridans as normal floras (Table 1). Escherichia coli accounted for the highest number of isolates in both the healthy and sick patients, followed by Klebsiella pneumoniae and Staphylococcus aureus. Thus these organisms were the main pathogens in this study. Candida albicans, Corynebacterium diphtheria and Lactobacillus were isolated only in the sick patients (Table 2).
Table 3 shows the list of chewing sticks used for this study. The pH of the chewing sticks extracts was determined. Normal saline solution was used as control for anti-microbial activity. There was no antimicrobial effect at low concentration of chewing sticks extracts. None of the 10% chewing stick extracts showed activity against any of the organisms, however, of the 50% chewing stick extracts, N. latifolia showed activity on S. aureus, B. catarrhalis an C. albicans. J. curcas demonstrated activity against E. coli, K. pneumoniae, S. aureus, B. catarrhalis and C. albicans. All the other chewing stick extracts did not show any activity on the organisms.Normal saline showed no antimicrobial effect. The effective pH ranged from 5.23 - 6.74 while the higher pH values did not show any antimicrobial activity (Table 4 and 5). The active principle of the extract might have inhibited the cell wall synthesis of the organism hence the zone of inhibition.
Thus extracts of N. latifolia and the sap of J. curcas demonstrated appreciable activity against aerobic Staphylococci. Studies have demonstrated haemostatic properties of J. curcas (Odusote et al., 1999).
| Table 1: | Occurrence of microorganisms in healthy and sick patients |
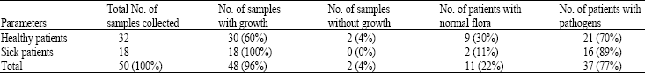 | |
| Table 2: | Organisms identified in healthy and sick patients |
 | |
| Table 3: | Identification of plant materials used |
 | |
| Table 4: | The activity of extracts of some chewing sticks and sap against oral pathogens |
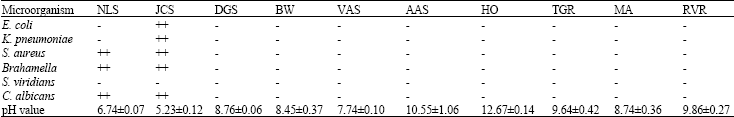 | |
| ++ = Sensitive organism - = Non-sensitive organism | |
| Table 5: | Diameter of zones of inhibition of micro organisms by extracts |
 | |
| ++ = Sensitive organism - = Non-sensitive organism | |
These extracts will provide cheap and affordable herbal mouthwash and toothpastes for HIV and other immunocompromised patients as well as anti-M ethicillin Resistant Staphylococcus aureus (MRSA) activity. Reports indicated that MRSA strains account for 10 to 40% of S. aureus isolated from some European hospitals (Voss et al., 1994). J. curcas is a shrub that grows wild in waste land all over the world. It is also a popular shrub for making hedges. It produces a viscid sap that drops in abundance from the injured leaf petioles or young tender parts of the stem. It is used for treating oral ulcers, candida patches on the oral mucosa and toothaches. N. latifolia also grows wild and is used as an antimalaria herb (Bouquet and Debray, 1974). Propagation of these plants is easy with the seeds. With the renewed interest in natural products that may have bio-prospecting values, these extracts will complement traditional hospital oral care protocol (chlorhexidine, hydrogen peroxide, sodium bicarbonate, thymol glycol, benzocaine mouth rinse and nystatin) in primary health care.