Research Article
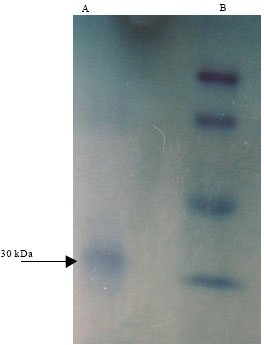
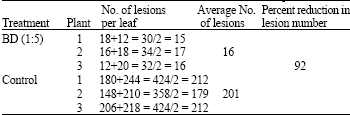
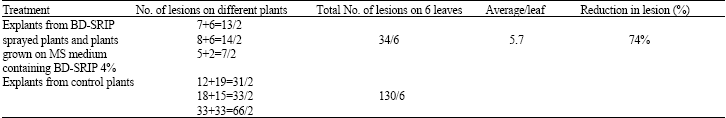
In vivo and in vitro Resistance Induction in Tobacco by Boerhaavia diffusa Systemic Resistance Inducing Protein and Transfer of Induced Resistance in in vitro Tobacco Plants
Biotechnology Division, Regional Research Laboratory, Sanat Nagar Srinagar, Jamu-Kashmir
Arif Jan
Biotechnology Division, Regional Research Laboratory, Sanat Nagar Srinagar, Jamu-Kashmir
H.N. Verma
Departement of Botany, Lucknow University, Lucknow, India