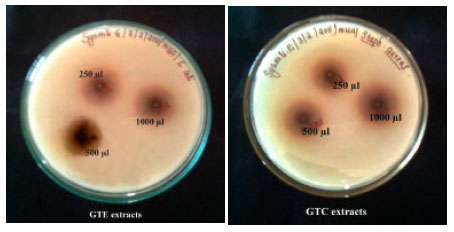
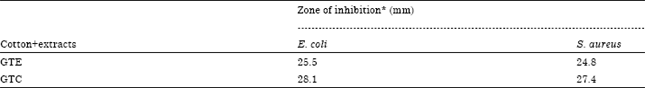
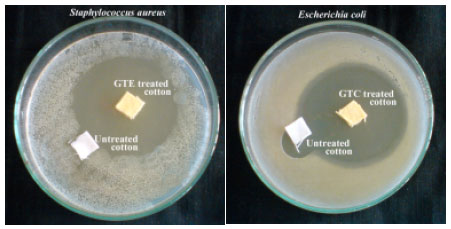
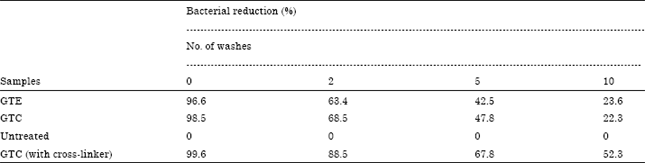
Research Article
Antibacterial Cotton Finish Using Green Tea Leaf Extracts Interacted with Copper
Department of Microbiology and Bioinformatics, CMS College of Science and Commerce, Coimbatore, India
B. Elayarajah
Department of Microbiology and Bioinformatics, CMS College of Science and Commerce, Coimbatore, India
Kulanthaivelu
Department of Microbiology, Karpagam University, Coimbatore
R. Rajendran
Post Graduate and Research Department of Microbiology, PSG College of Arts and Science, Coimbatore, India
B. Venkatrajah
Department of Microbiology and Bioinformatics, CMS College of Science and Commerce, Coimbatore, India
P. Ajith Kumar
Department of Microbiology and Bioinformatics, CMS College of Science and Commerce, Coimbatore, India