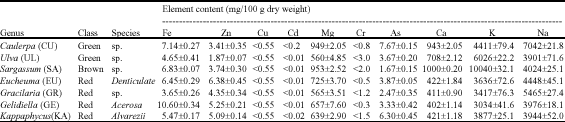
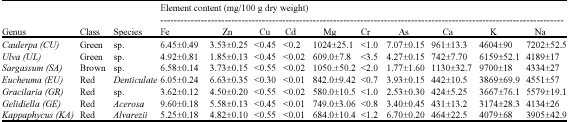
Research Article
Mineral Content of Some Seaweeds from Sabah’s South China Sea
School of Engineering and Information Technology, University Malaysia Sabah, 88999 Kota Kinabalu, Sabah, Malaysia
Rosalam Sarbatly
School of Engineering and Information Technology, University Malaysia Sabah, 88999 Kota Kinabalu, Sabah, Malaysia
D.M.R. Prasad
School of Engineering and Information Technology, University Malaysia Sabah, 88999 Kota Kinabalu, Sabah, Malaysia
Awang Bono
School of Engineering and Information Technology, University Malaysia Sabah, 88999 Kota Kinabalu, Sabah, Malaysia