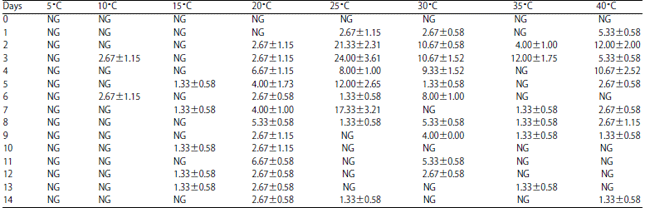
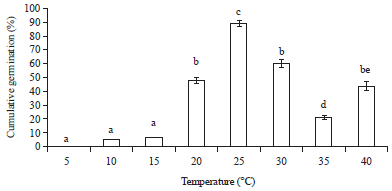
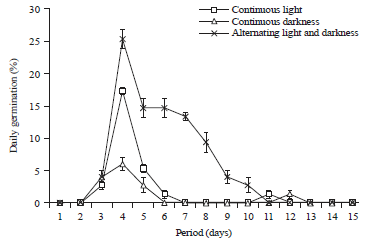
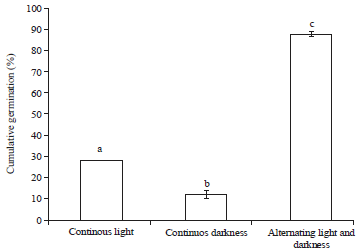
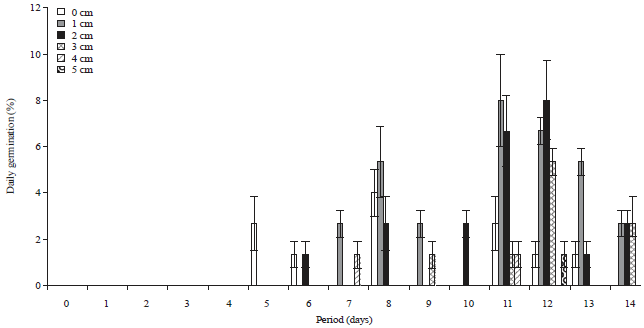
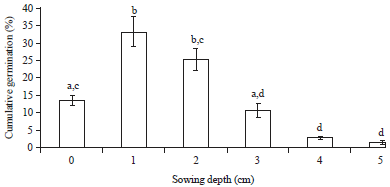
Research Article
Effect of Temperature, Light and Sowing Depth on Seed Germination of Celosia argentea L.
Medicinal Plants and Economic Development (MPED) Research Centre, Department of Botany, University of Fort Hare, Alice 5700, South Africa
LiveDNA: 234.20399
Gloria Aderonke Otunola
Medicinal Plants and Economic Development (MPED) Research Centre, Department of Botany, University of Fort Hare, Alice 5700, South Africa
LiveDNA: 234.16249
Anthony Jide Afolayan
Medicinal Plants and Economic Development (MPED) Research Centre, Department of Botany, University of Fort Hare, Alice 5700, South Africa