Review Article
In vitro Selection: A Candidate Approach for Disease Resistance Breeding in Fruit Crops
Biotechnology Lab, Central Institute for Subtropical Horticulture, Rehmankhera, Lucknow-227107, India
Madhu Kamle
Biotechnology Lab, Central Institute for Subtropical Horticulture, Rehmankhera, Lucknow-227107, India
Anju Bajpai
Biotechnology Lab, Central Institute for Subtropical Horticulture, Rehmankhera, Lucknow-227107, India
M. Muthukumar
Biotechnology Lab, Central Institute for Subtropical Horticulture, Rehmankhera, Lucknow-227107, India
Shahina Kalim
Department of Biochemistry, Bundelkhand University, Jhansi-284128, India










pradeep kumar Reply
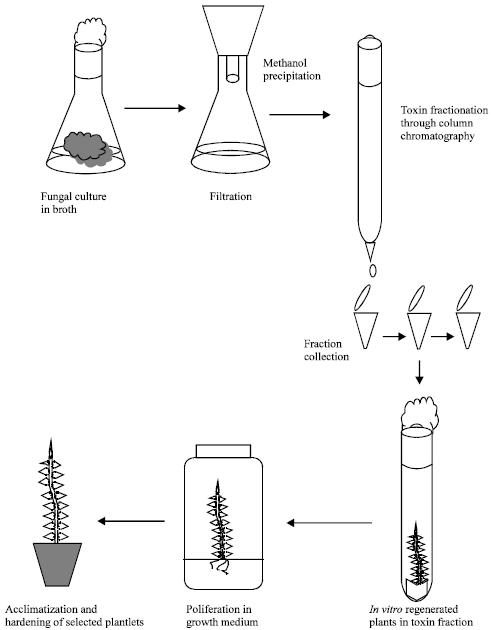
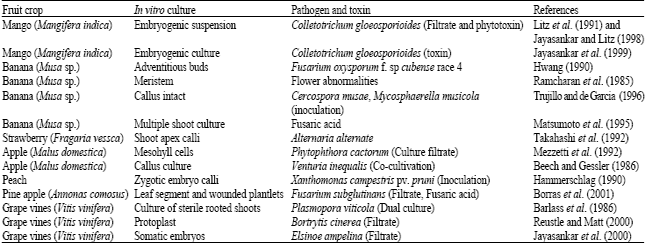
Very good paper with all information regarding "in vitro selection" and also related to somaclonal variation study in relation to resistant development by using toxin..