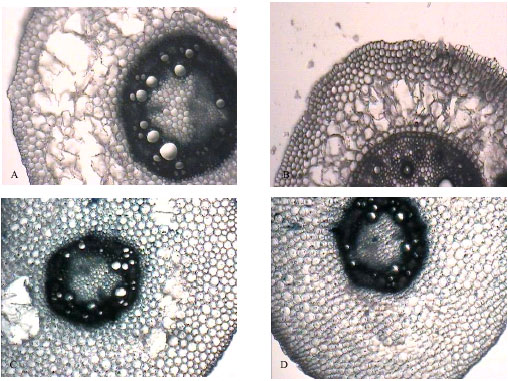
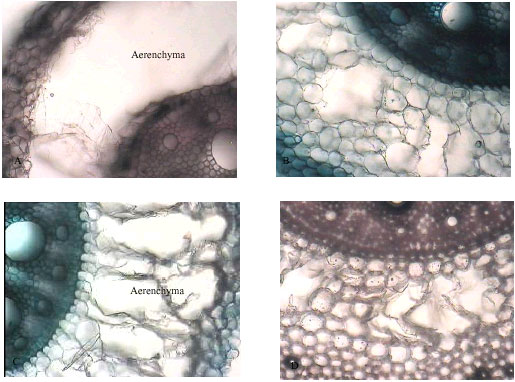
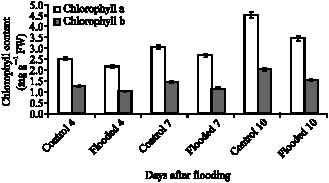
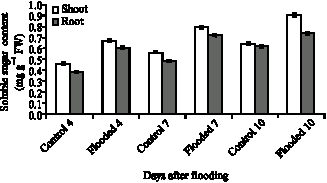
Research Article
Effects of Three Different Flooding Periods on Some Anatomical, Morphological and Biochemical Changings in Maize (Zea mays L.) Seedlings
Department of Biology, Urmia University, Urmia, Iran
R. Heidary
Department of Biology, Urmia University, Urmia, Iran
T. Farboodnia
Department of Biology, Urmia University, Urmia, Iran