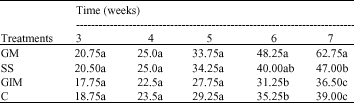
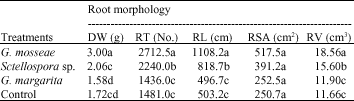
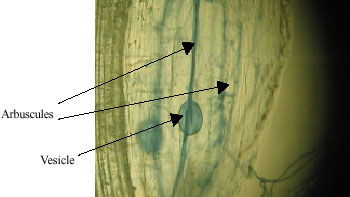
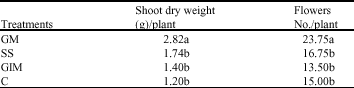Research Article
Response of (Lycopersicum esculentum Mill.) to Different Arbuscular Mycorrhizal Fungi Species
Faculty of Agriculture, Universiti Putra Malaysia 43400 UPM,Serdang, Selangor Darul Ehsan, Malaysia
S. Kamaruzaman
Faculty of Agriculture, Universiti Putra Malaysia 43400 UPM,Serdang, Selangor Darul Ehsan, Malaysia
O. Radziah
Faculty of Agriculture, Universiti Putra Malaysia 43400 UPM,Serdang, Selangor Darul Ehsan, Malaysia
J. Kadir
Faculty of Agriculture, Universiti Putra Malaysia 43400 UPM,Serdang, Selangor Darul Ehsan, Malaysia
H.N. Masdek
Malaysian Agricultural Research and Development Institute, Malaysia