Research Article
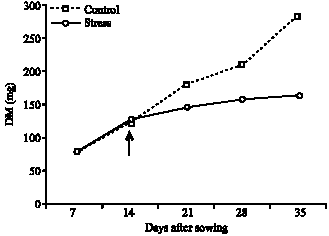
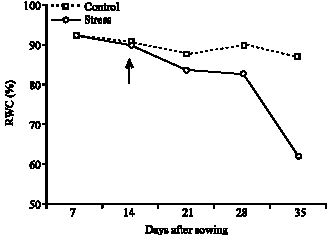
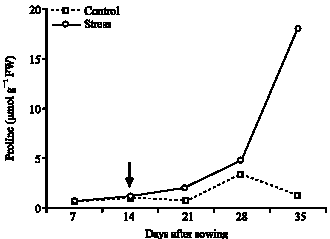
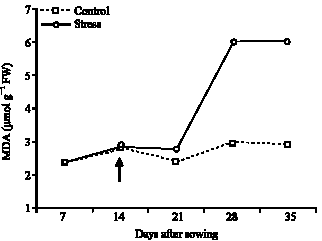
Influence of Water Stress on Proline Accumulation, Lipid Peroxidation and Water Content of Wheat
Department of Field Crops, Faculty of Agriculture, Ege University, Bornova, Izmir, Turkey
Mithat Nuri Gevrek
Department of Field Crops, Faculty of Agriculture, Ege University, Bornova, Izmir, Turkey