ABSTRACT
Germination percentage, seedling vigour, lipid composition and carbohydrate composition and its related enzymes were determined in cotyledons of soybean during storage for 180 days. Percent germination and vigour index of soybean seeds decreased with storage period for 180 days and seeds stored either in polythene or jute bags at 15°C showed higher germination potential and vigour index in comparison to those stored at room temperature. The dry matter content of cotyledons from germinating soybean seeds stored under different storage conditions increased with the increase in storage period. The total lipid content decreased initially up to 120 DOS and slightly increased thereafter, irrespective of the different storage conditions whereas proportion of free fatty acid, sterol and phospholipid content increased with a corresponding decrease in triglycerides content. Starch content in cotyledons declined from 90 to 180 DOS in all the treatments whereas the total soluble sugars, sucrose and reducing sugar content decreased up to 90 days of storage. The activity of α-amylase decreased up to 90 DOS and gradually increased thereafter, irrespective of the packing conditions and storage temperature. The invertase activity after germination decreased up to 90 DOS in seeds stored at RT and up to 120 DOS in seeds stored at 15°C. The data suggested that seeds stored at 15°C in both the packings maintain their viability up to 90 days of storage.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2007.502.507
URL: https://scialert.net/abstract/?doi=ajps.2007.502.507
INTRODUCTION
One of the major constraints in soybean cultivation is the non-availability of high vigour seeds at the time of sowing. Soybean seeds undergo rapid loss of vigour and viability during storage, which is more pronounced under sub-tropical conditions. Seed longevity is greatly influenced by storage conditions, such as relative humidity and temperature and lowering of these parameters significantly increases the storage life of seeds (Sauer et al., 1992). Deterioration of seed during storage is manifested as a reduction in percent germination while those seeds that do germinate, produce weak seedlings, which ultimately affect the growth and yield of crop plant (Tekrony et al., 1993). Contents of soluble carbohydrates generally decline with aging of seed (Petruzelli and Taranto, 1989; Sharma et al., 2005) and this decline might result in limited availability of respiratory substrates for germination. Depletion of disaccharides may lessen the protective effects of sugars on structural integrity of membrane (Crowe et al., 1984). The lipid related changes of seeds during storage revealed decline in phospholipids and polyunsaturated fatty acids leading to marked decline in seed vigour (Priestley and Leopold, 1983). An increase in necrosis in cotyledons and substantial reduction in total germination was reported in soybean seeds stored at high temperature by Falivene et al. (1980). Somchai (1999) reported decreased percentage of seed germination and seedling vigour with increase in storage period. Soybean seeds stored at controlled temperature (15-20°C) had a higher percentage germination than those stored at ambient temperature (Somchai, 1999; Sharma et al., 2006). Earlier work from our laboratory revealed that the total soluble sugars and sucrose contents increased while the reducing sugar contents of seeds decreased during its storage in polythene or jute bags (Sharma et al., 2005) so development of appropriate storage conditions can ensure a satisfactory level of seed quality at the time of planting. The present investigation reports the biochemical changes in cotyledons of germinating soybean seeds as influenced by different storage conditions.
MATERIALS AND METHODS
Soybean seeds of Cv. PK 416 were obtained from department of Plant Breeding, Genetics and Biotechnology, Punjab Agricultural University, Ludhiana.
The seeds were sun dried to about 9% moisture level and placed in polythene or jute bags in storage at room temperature and 15°C (BOD incubator) for a period of 6 months. Seed samples were drawn at an interval of 30 days. Seeds were surface sterilized with 0.1% mercuric chloride, soaked in sterile distilled water for 6 h and were allowed to germinate on wet paper rolls in dark at 25°C in the BOD incubator. The percentage germination was calculated out of total seeds subjected to germination. Vigour index was expressed as germination percentage multiplied by the length of the axis measured after 7 days of germination. The cotyledons were separated from the embryonic axis and a weighed quantity of tissue was oven dried at 60°C for 48 h to constant weight. After drying, the tissue was immediately placed in a desiccator before the final weighing. A portion of the cotyledonary tissue was crushed and boiled in a few ml of isopropanol to inactivate phospholipases, then homogenized in 20 volumes (w/v) of chloroform and methanol (2:1 v/v) and stored at 0-4°C until used. A portion of the tissue was put into boiling 80% ethanol and stored. The extraction of lipids was done by the method of Folch et al. (1957). A suitable aliquot of lipid extract was evaporated to dryness to determine the lipid content by weighing. The methods used for the estimation of different lipid classes were according to Munshi et al. (1990). The extraction and estimation of starch, total soluble sugars and reducing sugars were done by earlier described methods (Munshi et al., 1990).
For preparation of various enzyme extracts, 1 g of cotyledonary tissue was homogenized in a pre-chilled pestle and mortar by freezing and thawing in 10 volumes of extraction medium (0.2 M sodium acetate buffer, pH 4.8 containing 1% polyvinylpyrrolidine for invertase; 0.1 M sodium acetate buffer pH 6.0 containing 0.2% calcium acetate for α-amylase and 0.1 M sodium acetate buffer pH 3.6 containing 0.1 mM EDTA and 1% papain for β-amylase). The homogenate was filtered through double layers of cheese cloth and centrifuged at 20,000 g for 30 min. The lipid cake was removed and the supernatant was again centrifuged. The aqueous phase between the organelle pellet and newly formed cake was separated and termed as crude enzyme extract. The activities of α- and β-amylases (Duffus and Rosie, 1973) and invertase (Singh et al., 1978) were determined in these extracts. The protein content was determined by the method of Lowry et al. (1951). The experiment was performed in duplicate with three replications and data was analyzed statistically by using ANOVA test for determining the critical differences between different groups.
RESULTS AND DISCUSSION
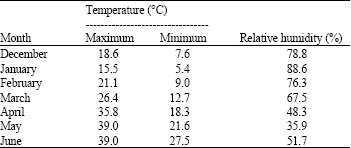
The data regarding the room temperature and relative humidity during the period of storage has been given in Table 1. Germination studies are important for determining the quality of the seeds for raising the crop. Soybean seeds collected at harvest showed 89% germination at 7th day of germination and it declined during storage in all the treatments. The seeds stored at 15°C in both the packings showed higher germination percentage than those stored at RT and maintained germinability above minimum standard of certification ie up t o 70% for 120 days of storage. Seedling vigour index were also declined with storage of seeds and the seeds stored at 15°C showed better vigour index in comparison to those at rmoom temperature (Fig. 1). The higher percent germination values at 0 DOS might be due to the higher amounts of soluble sugars present in the seeds (Sharma et al., 2006) and that reflects corresponding high vigour index and germinability of the seeds. Edje and Burris (1970) have opined that decline in soluble sugars might result in limited availability of respiratory substrate for germination.
| Table 1: | Temperature and relative humidity during December 2002 to July 2003 at Punjab Agricultural University, Ludhiana |
 | |
 | |
| Fig. 1: | Germination percentage and vigour index of germinating soybean seeds under different storage conditions. Each point represents the mean of three replications. RT- room temperature |
 | |
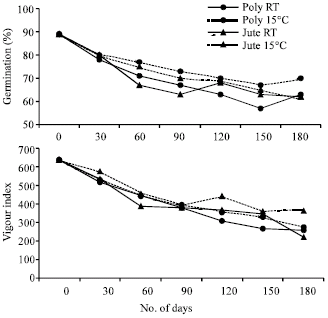
| Fig. 2: | Changes in dry matter and lipid content in cotyledons of germinating soybean seeds during storage. Each point represents the mean of three replications. CD (p>0.05) for DOSx PMxT is 0.19 for lipid content. (DOS: days of storage, PM: packing material and T: temperature) |
Loss in seedling vigour is reported to precede the loss of seed viability in number of crops including soybean (Dharamlingam and Basu, 1990). Similar results of decreased germination percentage and seedling vigour index with aging were reported by Singh and Dadlani (2003) and Gupta and Aneja (2004) in soybean. The decrease in % germination and vigour index can be attributed to DNA degradation with aging, which leads to impaired transcription causing incomplete, or faulty enzyme synthesis essential for earlier stages of germination.
The dry matter content of cotyledons from germinating soybean seeds stored under different conditions increased with the increase in storage period up to 180 days (Fig. 2) and increase in dry matter content in cotyledons of germinating soybean seeds stored in jute bags at 15°C or RT was more as compared to those stored in polythene bags beyond 120 DOS. The total lipid content decreased initially up to 120 DOS in all the treatments and slightly increased thereafter in seeds stored in polythene bags at RT or 15°C (Fig. 2).
| Table 2: | Changes in lipid composition in cotyledons of germinating soybean seeds during storage |
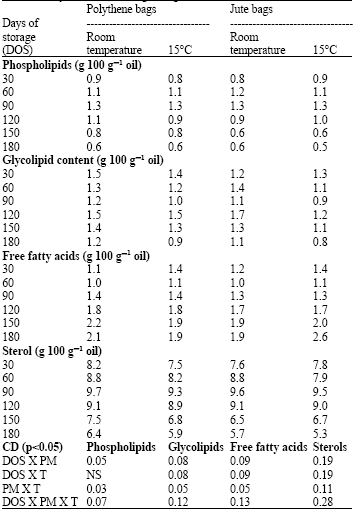 | |
The values are procured on the 7th day of germination. PM-packing material; T-temperature; NS-nonsignificant, Values at zero DOS: PL-0.71, GL-1.41, FFA-1.5 and Sterol-6.6 g 100 g-1 oil.
The phospholipid content increased up to 90 DOS followed by a decrease with further increase in storage period up to 180 DOS, irrespective of the packing and temperature conditions (Table 2). The glycolipid content did not vary much in germinating seeds stored in polythene or jute bags with the storage period however, the decrease in glycolipid content was more at 15°C in both the packings as compared to RT. On germination, the free fatty acid content in cotyledons of germinating soybean seeds increased with the increase in storage period from 30 to 180 days in all the treatments. The sterol content increased up to 90 DOS and then decreased with the increase in storage period irrespective of the packings and temperature conditions. The increase in sterol content during the initial 90 days of storage was notably more in the seeds stored at RT than those stored at 15°C in polythene bags.
In the present studies, the data on lipid composition has revealed that lipid degradation marginally decreased triglyceride content during storage and increased free fatty acids, sterol and phospholipid content suggesting that the lipase present in the seeds remains active and alters membrane integrity (Bernal Lugo and Leopold, 1992). The damage to membrane integrity has also been indicated by the decrease in sucrose content during initial period of storage (Fig. 3).
The starch content in the germinating cotyledons showed a significant decrease from 90 to 180 DOS in all the treatments (Fig. 3). The total soluble sugar content decreased from maturity to 90 DOS in all the treatments followed by an increase up to 150 DOS and this decrease in total soluble sugar content was more in seeds stored in polythene bags at RT or 15°C as compared to those stored in jute bags. The sucrose content decreased up to 90 DOS irrespective of the packings and temperature conditions (Fig. 3) and increased with further increase in storage period up to 180 days, at RT while there was a decline in the sucrose content in germinating cotyledons with increase in storage period beyond 150 days at 15°C. The reducing sugar content of cotyledons showed a marked decline up to 90 DOS from an initial high value at maturity followed by accumulation of reducing sugars at 120 DOS irrespective of packing conditions and storage temperature used. A further decrease was observed in the reducing sugar content of germinating soybean seeds at 150 and 180 DOS, which might be caused by their utilization for respiration.
The activity of β-amylases in the germinating cotyledons of soybean seeds decreased nonsignificantly initially up to 120 DOS and increased on further increase in the storage period in seeds stored in polythene bags at RT or 15°C (Fig. 4). The α-amylase activity decreased up to 90 DOS cotyledons followed by a gradual increase in its activity from 90 to 120 DOS in all the treatments. The invertase activity decreased up to 90 DOS when the seeds were stored at room temperature in both jute and polythene bags. However, there was decline in invertase activity up to 120 DOS in seeds stored in both the packings at 15°C. Increase in storage period resulted in enhancement of invertase activity in all the treatments up to 180 DOS.
In the present studies the decrease in total soluble sugars during initial period of storage up to 90 DOS suggested that carbohydrates in the cotyledons are the primary constituents utilized during germination before seedling growth commences (Rosenberg and Rinne, 1987). Adams et al. (1980) reported that the total soluble sugars are rapidly utilized in germinated soybean cotyledons, declining to about 20% of their initial level. The decrease in total soluble sugars might be due to their utilization in respiration (Meyer and Mayber, 1966), incorporation into cell walls or translocation to the growing embryo (Meyer and Anderson, 1952).
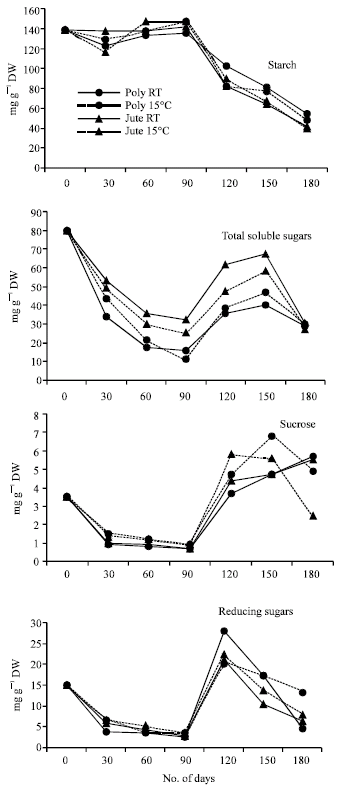 | |
| Fig. 3: | Changes in carbohydrate composition in cotyledons of germinating soybean seeds during storage. Each point represents the mean of three replications. CD (p>0.05) for DOSx PMxT is 1.30 for starch; 0.78 for total soluble sugars; 0.41 for reducing sugars and 0.17 for sucrose (DOS: days of storage, PM: packing material and T: temperature) |
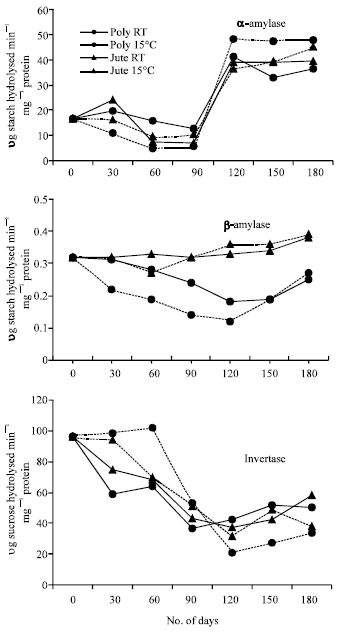 | |
| Fig. 4: | Changes in amylases and invertase activities in cotyledons of germinating soybean seeds during storage. CD (p>0.05) for DOSx PmxT is 1.75 for α-amylase; 0.02 for β-amylase and 1.33 for invertase (DOS: days of storage, PM: packing material and T: Temperature) |
The increase of total soluble sugars beyond 90 DOS might be due to enhanced starch hydrolysis by a corresponding increase in α-amylase activity during storage in all the treatments (Fig. 3 and 4). The presence of starch and the corresponding enzymes for its hydrolysis have already been well documented in soybean seeds (Wilson et al., 1978; Hildebrand and Hymowitz, 1981).
The depletion in the sucrose content up to 90 DOS might lessen the protective effects of sugars on structural integrity of the membranes which ultimately affect the ability of seeds to maintain the vitrified state, a non-crystalline liquid state of high viscosity (Williams and Leopold, 1991). The accumulation of sucrose beyond 90 DOS parallels the increase in total soluble sugars of germinating seeds which might be caused by the degradation of oligosaccharides during this period and also due to decrease in the invertase activity (Fig. 4).
The increase in the starch content of the seeds after germination as compared to mature seeds (Sharma et al., 2006) support the earlier observations of a number of workers (Webster and Leopold, 1977) that soybean seeds produce starch during imbibition and germination which is a transient reserve material for germinating soybean cotyledons (Adams et al., 1980). Probably the newly formed starch is produced by gluconeogenesis using precursors from the oil reserves. The higher values of starch content up to 90 DOS may be due to the decline in the α-amylase activity in the cotyledons of seed stored under different conditions. However, the increased α and β-amylase activities beyond 90 DOS correspond with the decrease in the starch content and increased amount of total soluble sugars in cotyledons of germinating soybean seeds. The majority of total soluble sugars, which is sucrose and the increase in sucrose and reducing sugar content beyond 90 DOS, might be due to hydrolysis of raffinose oligosaccharides during storage.
The overall perusal of data suggests that soybean seeds undergo compositional changes during period of storage. Seeds stored in polythene or jute bags at RT or 15°C after harvest undergo sugar hydrolysis as shown by decreased total soluble sugar, reducing sugar and sucrose that affected the seed viability and vigour of the seedling. Total lipids decreased with an increase in phospholipids, free fatty acid and sterol content suggesting that gluconeogenesis is active in early phase of germination. The higher percent germination and vigour index of seeds stored at 15°C suggested that the storage of seeds at low temperature retained their vigour for longer period as compared to room temperature.
REFERENCES
- Adams, C.A., R.W. Rinne and M.C. Fjerstad, 1980. Starch deposition and carbohydrase activities in developing and germinating soybean seeds. Ann. Bot., 45: 577-582.
Direct Link - Bernal, L.I. and A.C. Leopold, 1992. Changes in soluble carbohydrates during seed storage. Plant Physiol., 98: 1207-1210.
Direct Link - Crowe, L.M., R. Mourdian, J.H. Crowe, S.A. Jackson and C. Womersly, 1984. Effects of carbohydrates on membrane stability at lower water activities. Biochem. Biophys. Acta, 769: 141-150.
CrossRef - Folch, J., M. Lees and G.H.S. Stanley, 1957. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem., 226: 497-509.
CrossRefPubMedDirect Link - Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem., 193: 265-275.
CrossRefPubMedDirect Link - Petruzelli, L. and G. Taranto, 1989. Wheat ageing: The contribution of embryonic and non-embryonic lesions to loss of seed viability. Physiol. Plant., 76: 289-294.
CrossRef - Priestley, D.A. and A.C. Leopold, 1983. Lipid changes during natural aging of soybean seeds. Physiol. Plant, 59: 467-470.
CrossRef - Rosenberg, L.A. and R.W. Rinne, 1987. Changes in seed constituents during germination and seedling growth of precociously matured soybean seeds (Glycine max L). Ann. Bot., 60: 705-712.
Direct Link - Singh, M.B., C.P. Malik and N. Thapar, 1978. Changes in the activities of some enzymes of carbohydrate metabolism in Amaryllis vittata pollen suspension cultures. Plant Cell Physiol., 19: 677-684.
Direct Link - Tekrony, D.M., C. Nelson, D.B. Egli and G.M. White, 1993. Predicting soybean seed germination during warehouse storage. Seed Sci. Technol., 21: 127-137.
Direct Link








