Research Article
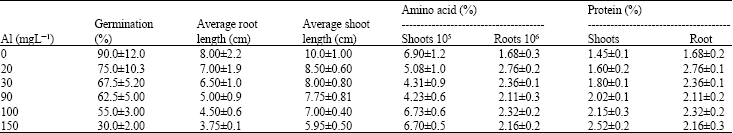
Aluminium Stress Induced Alteration in Seedling Growth and Alleviation in Protein and Amino Acid Contents of Lens culinaris
Department of Chemistry, Jinnah University for Women, 5C Nazimabad, 74600-Karachi, Pakistan
Sehrish Hasan
Department of Chemistry, Jinnah University for Women, 5C Nazimabad, 74600-Karachi, Pakistan
Fahim Uddin
Department of Chemistry, University of Karachi, Karachi, Pakistan