ABSTRACT
Seeds of two cultivars (Pusa chetki and Japanese white) of radish were subjected to the accelerated ageing at 40°C and 100% RH for various time intervals (0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 days). Accelerated ageing resulted in progressive loss of seed viability and vigour in both cultivars. A marked genotypic difference of seeds on ageing treatment had been recorded. Among two cultivars, seeds of Pusa chetki have been found to be more sensitive to ageing compared to those of Japanese white. Seeds of both cultivars had an initial cent percent germination which declined gradually to no germination after 9 days of ageing. In addition to seed germination, various other physiological parameters were recorded to assess the effect of ageing. Ageing led to decline in seed vigour in both cases. Sensitivity of seeds of Pusa chetki to ageing treatment had been substantiated by the Electrical Conductance (EC) of seed leachet, which elicited more increased value in this cultivar, compared to the other one (3.9 fold increase over a 2.5 fold increase in latter). Likewise, moisture content of seeds of Pusa chetki increased 4.2 fold against 3.8 fold in Japanese white, over the original moisture content in 10 days aged seeds. Total protein content declined in both cases following ageing treatments.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.461.464
URL: https://scialert.net/abstract/?doi=ajps.2006.461.464
INTRODUCTION
The ageing is an universal phenomena occurring in all living organisms during the natural course of development, however, unfavorable/stress conditions hastens it. Seeds of all plants exhibit a maximum potential for germination immediately after the harvest, which declines gradually with an increased storage period. Seed ageing is one of the key factors responsible for the decline in the yield of various food crops particularly the vegetables. Ageing of seeds is evident through various parameters viz., delayed germination and emergence, slow growth, increased susceptibility to environmental stresses (Walters, 1998). Many processes have been suggested as possible mechanisms involved in the seed deterioration like chromosomal damage, loss of activity of various metabolic enzymes, loss of ATP production/storage capacity, deterioration of membranes (Priestley, 1986). Changes in the metabolic activities associated with seed deterioration are highly complex and poorly understood (McDonald, 1999). Accelerated ageing techniques have great potential for understanding the mechanism of ageing and associated deterioration processes of seeds. The process of deterioration under accelerated ageing conditions are essentially similar to those under normal conditions, however, the major difference is that the rate of deterioration is much faster, thus, making it possible to study within reasonable time frame. A number of studies have been carried out in past to analyze the physiological and biochemical changes associated with accelerated aged seeds (Aiazzi et al., 1996; Goel and Sheoran, 2003; Vieira et al., 2004).
Radish is an important vegetable crop used throughout India. The present investigation aims to elucidate some of the physiological changes associated with ageing employing two cultivars of radish as well as to study the effect of genotypes on the ageing.
MATERIALS AND METHODS
Plant material: Healthy, infection free and uniform size seeds of two cultivars (Pusa chetki and Japanese white) of radish (Raphanus sativus L.) were used for all experiments. Certified seeds of these cultivars were procured from the National Seed Cooperation, Pusa Campus, India.
Accelerated ageing: For accelerated ageing, the seeds were exposed to a temperature of 40°C and 100% RH (Byrd and Delouche, 1971) for various time intervals (1, 2, 3, 4, 5, 6, 7, 8, 9, 10 days). Seeds which were not exposed to the ageing treatments were referred as '0 day'. After each treatment, seeds were bench dried at room temperature, sealed in the aluminum seed pouches and stored at 4°C till the experimental analyses. The experimental studies were carried out at National Bureau of Plant Genetics Resources, New Delhi.
Germination: Twenty five seeds from each treatment were placed on moistened two-layered germination paper. Seeds were placed in between two layers of germination papers and rolled. The rolled sandwich was further wrapped in butter paper and incubated in the seed incubators maintained at 25±2°C for seven days. Seeds with 0.5 cm radicle and plumule were considered as germinated (Schoettle and Leopold, 1984). Total number of germinated seeds, the shoot length and root length of seedlings were recorded. Vigour index was expressed as the product of percentage germination and the seedling length.
Imbibition: Twenty five pre-weighed seeds were placed in Petri plates lined with moist filter paper (Whatman No. 1). Seeds were left undisturbed for overnight and were weighed again. Percent imbibition was expressed as increase in weight over the initial fresh weights.
Moisture content of seeds: Fifteen seeds were taken in the moisture bottles. The initial weight (W1) of the empty bottle was taken. Seeds were crushed with pestle and mortar and placed in the moisture bottles and the weight was recorded (W2). Bottles were kept in the oven maintained at 105°C for overnight. The bottles were weighed again (W3). The percentage moisture was calculated as:
Percent moisture = (W2-W3/W2-W1) x 100 |
Electrical conductance: Twenty five seeds were weighed and placed in 100 mL beaker containing 30 mL distilled water. Beakers were covered with aluminum foil and left undisturbed for overnight. The elute was collected by decantation and the final volume was made to 50 mL with distilled water (Simon and Rajaharun, 1972). The electrical conductance was measured and expressed as μS mg-1 dry wt (Stewart and Bewley, 1980).
Total protein content: One hundred milligram of seeds were crushed in pre-chilled pestle and mortar using 5 mL of water. The extract was centrifuged at 10000 rpm for 15 min to collect the clear supernatant. The supernatant was used to quantify the protein content employing the method described by Lowry’s et al. (1951).
Statistical analysis of data: All experiments were carried out in three replicates maintaining the same number of seeds of both the varieties and all experiments were repeated twice. Data have been expressed as mean ± standard error. Data were subjected to analysis of variance (ANOVA) by Fischer’s least significant difference test (LSD) at the 5% level of significance (p≤0.05). Data expressed as percentage responses were arcsin transformed before analysis.
RESULTS AND DISCUSSION
Accelerated ageing results in the progressive loss of seed viability and vigour, which is evident through the results of the present study. Seeds of both cultivars of radish exhibited an initial 100% germination which declined to no germination after 9 days of ageing. Among two cultivars, seeds of Pusa chetki exhibited more susceptibility to the ageing over the seeds of Japanese winter as at any time point, the percentage germination was less in the former case (Fig. 1). Besides, percentage germination, root length and shoot length also elicited a significant decline over the control seeds. The decline in all these parameters contributed to the decline in the seed vigour index in both cultivars. Seeds of cultivar Pusa chetki were found to be more affected compared to the other one (Table 1 and 2). These observations in decline in seed vigour are in accordance with the earlier works on Atriplex cordobensis (Aiazzi et al., 1996), Cajanus cajan (Kalpana and Madhav Rao, 1995).
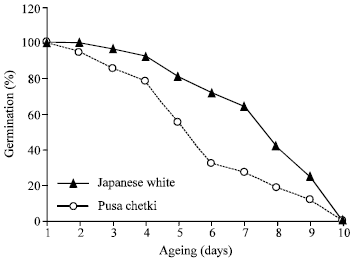 | |
| Fig. 1: | Comparison of percentage of germination of the seeds of two cultivars of radish when aged for different time durations |
| Table 1: | Physiological and biochemical changes in the accelerated aged seeds of radish cultivar, Pusa chetki |
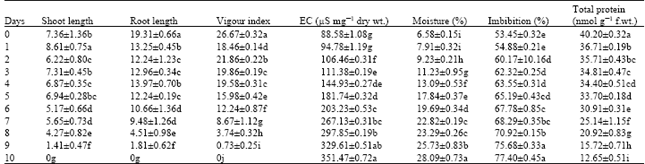 | |
| Values represent mean±Standard error. Values followed by same letter are not statistically significant from each other (p≤0.05) | |
| Table 2: | Physiological and biochemical changes in the accelerated aged seeds of radish cultivar, Japanese white |
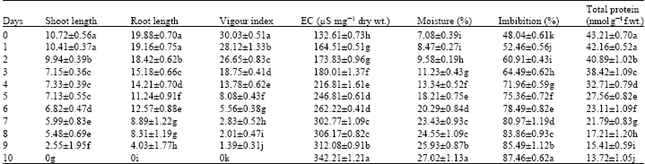 | |
| Values represent mean±Standard error. Values followed by same letter are not statistically significant from each other (p≤0.05) | |
Differential effect of genotypes to sustain the ageing treatments has also been reported earlier (Anderson and Gupta, 1986, Filho et al., 2001). Likewise, differential response of genotypes to withstand the ageing has been reported for naturally aged seeds of cotton (Goel and Sheoran, 2003).
The electrical conductance test of seed leachets measures the amount of electrolytes that are leaked into the imbibing medium from seeds. This test reflects the integrity of cell membrane subsequent to the treatment. Our observations of electrical conductance in both cultivars revealed a gradual increase in the values from 88.58±1.08 to 351.47±0.72 and 132.61±0.73 to 342.21±1.21 μS mg-1 dry wt. for Pusa chteki and Japanese white, respectively, from control seeds to 10 days aged seeds. The increased electrolyte leakage can be correlated with the decreased vigour index in seeds of both cultivars following ageing treatments (Table 1 and 2). Chang and Sung (1998) reported that the ageing treatment caused inability of seeds to maintain the integrity of membranes and this accounts for the reduced germinability of seeds. Kalpana and MadhavRao (1995) from their studies on Cajanus cajan reported that damage to the membranes. However in the aged seeds it is beyond repair and this accounts for the increased permeability of membranes, however, since the content of various leaked solutes is not uniformly increased indicates that the semi permeability nature of membranes is not affected by the ageing treatments. Increased conductance of the leachet of aged seeds has been documented earlier (Pérez and Arguello 1995; Goel and Sheoran, 2003; Vieira et al., 2004).
The moisture content of seeds increased with the ageing of seeds in both cultivars of radish. This increase could be explained by the disorganization of the cell membranes. Due to this reason the percentage imbibed water increased with the ageing (Table 1 and 2).
The total protein content of aged seeds decreased progressively with the number of days of treatments over the control seeds in both cultivars (Table 1 and 2). This decline could be attributed to either decreased rate of synthesis of proteins or increased degradation of stored proteins due to increased activity of proteinases, or the combination of two. Both cultivars elicited almost the similar pattern of decline in the protein content in relation to the number of days of treatments. Cherry and Skadsen (1986) hypothesized that the irreversible loss of some essential proteins in the aged seeds leads to loss of seed viability. The decline in the total protein content due to impaired protein biosynthetic activity with the gradual loss of seed viability have been reported in seeds of rye (Hallam et al., 1973), pea (Bray and Chow, 1976), sal (Nautiyal et al., 1985) and pigeon pea (Kalpana and MadhavRao, 1997).
To conclude, though it is a preliminary study to examine the effect of ageing on the biochemical and physiology of seeds, but our results are consistent with the previous reports highlighting that the ageing causes disorganization of membrane integrity which ultimately leads to the decline in the seed viability. However, the extent of damage to the membranes depends on the genetic makeup of the species as two cultivars used in the present study exhibited a varied degree of susceptibility to these ageing treatments.
ACKNOWLEDGMENTS
The authors thank the Director, NBPGR, New Delhi, India, for providing the facilities. The financial support of NAT project is gratefully acknowledged.
REFERENCES
- Aiazzi, M.T., J.A. Arguello, A. Perez, J. Di Rienzo and C.A. Guzman, 1997. Deterioration in Atriplex cordobensis (Gandoger et Stuckert) seeds: Natural and accelerated ageing. Seed Sci. Technol., 25: 147-155.
Direct Link - Bray, C.M. and T.Y. Chow, 1976. Lesions in post ribosomal supernatant fractions associated with loss of viability in pea (Pisum arvense) seed. Biochem. Biophys. Acta, 442: 1-13.
Direct Link - Chang, S.M. and J.M. Sung, 1998. Deteriorative changes in primed sweet corn seeds during storage. Seed Sci. Technol., 26: 613-625.
Direct Link - Filho, J.M., A.D.C. Novembre and H.M.C.P. Chamma, 2001. Accelerated ageing and controlled deterioration seed vigour tests for soybean. Sci. Agric., 58: 421-426.
Direct Link - Goel, A. and I.S. Sheoran, 2003. Lipid peroxidation and peroxide-scavenging enzymes in cotton seeds under natural ageing. Biol. Plantarum, 46: 429-434.
CrossRefDirect Link - Hallam, N.D., B.E. Roberts and D.J. Osborne, 1973. Embryogenesis and germination in rye (Secale cereale L.): III. Fine structure and biochemistry of the non-viable embryo. Planta, 110: 279-290.
Direct Link - Kalpana, R. and K.V. MadhavRao, 1995. On the ageing mechanism in pigeonpea (Cajanus cajan (L.) Mill sp.) seeds. Seed Sci. Technol., 23: 1-9.
Direct Link - Kalpana, R. and K.V.M. Rao, 1997. Protein metabolism of seeds of pigeonpea (Cajanus cajan (L.) Mill sp.) cultivars during accelerated ageing. Seed Sci. Technol., 25: 271-279.
Direct Link - Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem., 193: 265-275.
CrossRefPubMedDirect Link - McDonald, M.B., 1999. Seed deterioration: Physiology, repair and assessment. Seed Sci. Technol., 27: 177-237.
Direct Link - Nautiyal, A.R., A.P. Thapliyal and A.N. Purohit, 1985. Seed viability in sal. 4. Protein changes accompanying loss of viability in Shorea robusta. Seed Sci. Technol., 13: 83-86.
Direct Link - Perez, M.A. and J.A. Arguello, 1995. Deterioration in peanut (Arachis hypogaea L. cv. Florman) seeds under natural and accelerated aging. Seed Sci. Technol., 23: 439-445.
Direct Link - Schoettle, A.W. and A.C. Leopold, 1984. Solute leakage from artificially aged soybean seeds after imbibition. Crop Sci., 24: 835-838.
Direct Link - Simon, E.W. and R.R. Harun, 1972. Leakage during seed imbibition. J. Exp. Bot., 23: 1076-1085.
CrossRefDirect Link - Stewart, R.R.C. and J.D. Bewley, 1980. Lipid peroxidation associated with accelerated ageing of soybean axes. Plant Physiol., 65: 245-248.
Direct Link - Vieira, R.D., A.S. Neto, R.M. deBittencourts and M. Panobianco, 2004. Electrical conductance of the seed soaking solution and soybean seedling emergence. Sci. Agric., 61: 164-168.
Direct Link - Walters, C., 1998. Understanding the mechanisms and kinetics of seed ageing. Seed Sci. Res., 8: 223-244.
Direct Link








