Research Article
The Effect of Cytokinin Type and Concentration on Multiplication Rate of Spathiphyllum (Fam. Araceae)
Not Available
Meltem Mazmanoglu
Not Available
Yesim Yalcin Mendi
Not Available
Sedat Serce
Not Available
Selim Cetiner
Spathiphyllum spp. are popular foliage plants because of with their attractive foliage and growth habit, white fragrant flowers and tolerance of interior environments[1]. Like in many other ornamentals, vegetative propagation is the major method in Spathiphyllum[2]. The major propagation material is lateral and adventitious shoots. Although this method is rapid and can be used regardless of season, the offspring propagated by this method are few and do not have the desired vigor in the winter time[3].
The tissue culture methods for Spathiphyllum are reported [3-6]. This method has several advantages such as rapid clonal multiplication, pathogen elimination, germplasm storage, facilitation of transport as well as dissemination of new plant material[7]. The rate at which cultures grow and multiply during micropropagation is effected by physical nature of medium[4]. Nutrient media, including their mineral constituents, carbohydrate content and qualitative and quantitative aspects of growth regulators play an important role in micropropagation[6].
Cytokinins are growth regulators promoting cell division and cell expansion in plant tissue culture. Applications of cytokinin in tissue culture are promotion of adventitious shoot formation, prevention of senescence, reversion of the effect of auxin on shoots and occasionally, inhibition of root formation. Many studies have reported suitable cytokinin types and their concentrations for several species[8]. For example, Jinshu[9] studied the effect of cytokinins in tissue culture of Spathiphyllum. They reported that among root and stem explants of Spathiphyllum sp. were cultured on MS medium supplemented with different hormone, MS medium with 2 mg BA-1 and 1 mg L-1 kinetin yielded the most favorable results for bud induction. They found that by adding 1 mg LBA-1 and 0.1 mg LIAA-1 was helpful for bud propagation.
Explant growth and shoot multiplication of Spathiphyllum and Syngonium were compared on agar solicited medium and interfacial membrane rafts floating on liquid medium. After 25 days culture, greater shoot multiplication and fresh mass gain were achieved by plant material grown on rafts[4]. More recently, Ramirez-Malagon et al.[5] reported micropropagation of Spathiphyllum using a combination of two cytokinins with three different concentration of auxins. In view of potential commercial value, it is highly desirable to develop a method for rapid, efficient and large scale multiplication of Spathiphyllum. Herein, we report the optimized micropropagation of Spathiphyllum from shoot tip culture.
Plant material: The Spathiphyllum Sweet Pablo which is a major cultivar in the tropical foliage industry. Young Spathiphyllum plants were obtained from a commercial nursery. In culturing of plant materials heavily sterilization procedures were used to control contaminants. The plants were washed thoroughly for 30 min under running tap water followed by removal of leaves and roots. Basal part of plants including shoot tip about 2.5 cm were excised. The explants were treated with 0.05% (w/v) mercuric chloride with some drops of Tween 20 as a detergent for 10 min followed by rinses with sterile distilled water three times and after removal of leaves at a length or width of 0.5 cm. The explants were rinsed in 70% ethanol and were then soaked 10% (v/v) NaOCl solution with few drops of Tween 20 solution for 10 min followed three times of sterile distilled water. This step was repeated twice. The material was laid on to the initiation medium.
Culture medium and conditions: The culture medium used was Murashige and Skoog[10] basal medium supplemented with two types of cytokinins (BA: SIGMA B3408; PBA: SIGMA B2275) with 3% (w/v) sucrose and 8% (w/v) agar. The growth regulators used were PBA (1 and 2 mg L-1) and BA (1 and 2 mg L-1), in addition, a control without growth regulators was included.
All media were adjusted to pH 5.7 with 0.1 N NaOH or HCl and were autoclaved at 1.05 kg cm-2 121°C for 15 min. Each 25 x 150 mm tube contained one explant on 10 ml of medium and covered with a Magenta 2 way cap. All cultures were incubated at 25±1°C with a 16/8 h light/dark photoperiod illuminated by a cool-white fluorescent light (50 μmol m-2 s-1).
Multiplication of shoot cultures: The mean multiplication rate was calculated as the number of shoots and buds per initial shoot at the end of each multiplication cycle (4 weeks). The length, fresh weight of shoots and the number of shoots suitable for rooting were also recorded.
Statistical analysis: Analyses of variance were conducted for a factorial design by GLM procedure of SAS using a factorial design. The means were separated by Tukey for subcultures and by Duncan for hormone treatments in each of the subculture treatments. The mean tables were constructed by using TABULATE procedure of SAS[11].
By use of the disinfestations method described above 90% of the explants were established successfully. This was accomplished by adding a 0.05% (w/v) mercuric chloride for 10 min.
Multiplication rate: No multiple shoots were initiated on MS medium devoid of plant growth regulator. But the development of single plants was the most favorable on the basal medium without hormone although multiple shoot initiation was observed in all the cytokinin treatments tested after 30 days in culture.
The highest multiplication rate was recovered at subculture 3 (mean = 55.59) and 4 (mean = 55.66) (Table 1). For these subcultures, BA 1 mg L-1 treatment resulted in values that are in the highest group (7.75 and 8.49 for subcultures 1 and 2, respectively).
The MS medium with BA 1 mg L-1 resulted in the most favorable multiple shoot initiation. At a continuous multiplication of Spathiphyllum on 1 mg L-1 BA media, the multiplication rate began to increase starting from 1st subculture, reached its maximum 4th subculture with 8.49 shoots/plant. Multiplication rate of Spathiphyllum on 2 mg L-1 PBA medium was the highest at 3rd subculture and thereafter started to decline (Table 1).
Shoot fresh weight: The average fresh weight of shoots is a factor to be considered for better growth and development of plants adapted to soil after in vitro propagation[5]. In the present experiment there were no significant differences in the 1st and 2nd subculture for the shoot fresh weight. The highest shoot weight were at subculture 4 (mean = 0.97) and at BA 1 mg L-1 treatment as well (1.17). The highest shoot fresh weight values were achieved when the explants were cultured supplemented with PBA (2 mg L-1) at 3rd subculture. Using of BA content with 1 mg L-1 resulted in highest shoots fresh weight at 4th and 5th subculture (Table 1).
Shoot length: Growth regulator concentration significantly influenced mean length of shoots. Adding cytokinins in the media led to an increase in the multiplication rate and to a decrease in shoot length. Different cytokinin treatments had no effect on shoot length because there was no high amount of cytokinin in plant tissue at first subculture. Therefore we obtained longer shoots in the control media. At continuous multiplication subculture the amount of accumulated cytokinin in plantlets caused to obtain small plant at 2nd, 3rd and 4th subculture.
| Table 1: | Means of multiplication rate, shoot weight and shoot length for Spathiphyllum Cv. Sweet Pablo cultured on the media supplemented with different hormone treatments for several subculture cycles |
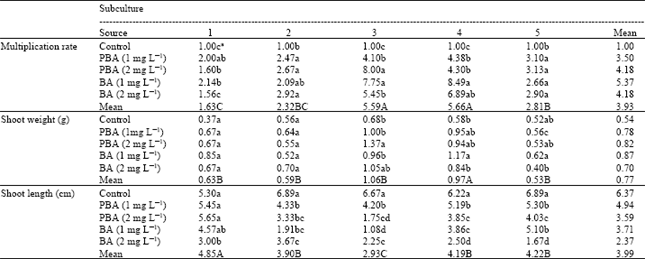 | |
| The capital letter(s) separate means for subcultures (row) while small letter(s) separate means for hormone treatments (columns) in each of the subculture treatments which were separated by Tukey and Duncan Tests, respectively. Shoot length data were recorded 4 weeks after transfer to multiplication medium | |
Using of PBA content to 1 mg L-1 starting from 2nd subculture to 5th subculture gave longer shoots after control. In the first subculture there is no significant differences between control, PBA (1 mg L-1) and PBA (2 mg L-1) for shoot length (Table 1).
The shoot length was lowest at subculture 3 (mean = 2.93) and at BA 1 mg L-1 treatment (1.08). Subculture 1 gave the longest shoot length (mean = 4.85) where the control had the largest mean (5.39) (Table 1).
Fonnesbech and Fonnesbech[3] had used 2 mg LBA-1 as cytokinin source in their study for the in vitro propagation of Spathiphyllum (Clevelandii). Spathiphyllum is an ornamental plant that is propagated by tissue culture around the globe; therefore, the cost of tissue culture is critical. PBA is about 30 times more expensive than BA, therefore has significant contribution in production costs. In this study, BA has been used as an alternative to PBA at 1 and 2 mg L-1 concentrations. Our results have revealed that BA has produced satisfactory number of shoots, but the quality of shoots was better with PBA.
The type of cytokinin applied had no major effect, both on the number of shoots produced and the fresh weight. On the other hand, the type of cytokinin applied had major effect on the quality of the shoots.
Varjda and Varjda[6] used BA in the Spathiphyllum multiplication medium. They observed 2 mg L-1 BA may be used in three successive subcultures as the 4th subculture this BA concentration led to a decrease in the number of high-grade shoot. Therefore, the content of BA was lowered to 0.5-1 mg L-1 for one or two subcultures. These findings is in accordance with present results. Especially in the first experiment both PBA and BA with two different concentrations starting from 4th and 5th subculture led to a decrease the number of shoots.
In conclusion, present study showed that at the end of fourth subculture adding PBA at 2 mg L-1 was most suitable for shoot formation and for obtained healthy and vigorous shoots.