ABSTRACT
Plants grown under saline conditions show changes in carbohydrate metabolism. Furthermore, calcium (Ca2+) plays an important role as a metabolic regulator. This work was carried out in order to assess the effect of external calcium application on sucrose metabolism and sugar contents in pepper plants (Capsicum annuum L. Cv. California) exposed to salinity. Calcium added with salinity showed ameliorative effect on growth and CO2 assimilation. In salinity-treated plants, the high hydrolytic activity observed could be a consequence of the activation of alternative pathways for obtaining energy. However, in plants treated with both salinity and calcium, the effect of calcium on sucrose phosphate synthase (SPS) and sucrose synthase (SS) could reveal an involvement (direct or indirect) by calcium. In any case, the effect of calcium application on sugar metabolism could explain its ameliorative effect under saline conditions.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.455.462
URL: https://scialert.net/abstract/?doi=ajps.2004.455.462
INTRODUCTION
The progressive salinization of land is considered as the major environmental factor limiting plant growth and productivity of the arid and semiarid regions. Salts that generally are common and necessary components of soil, being essential plant nutrients, can, at high levels, cause osmotic effects and nutrient ion imbalances that affect plant metabolism through repression or induction of several enzyme systems[1-3].
Plants adapt to stress by different mechanisms, including changes in morphological and developmental patterns as well as physiological and biochemical processes[4]. Adaptation to stress is associated with metabolic adjustments that lead to the accumulation of organic solutes such as sugars, polyols, betaines and proline[5].
Plant growth under high-salinity conditions can cause an excessive build-up of ions in leaf tissue leading to a reduction in the photosynthetic area of the plants as well as a decrease in CO2-fixation. These changes decrease the electron transfer in photosystem II, thus affecting sugar distribution, accumulation and mobilisation and giving rise to a carbohydrate production insufficient to support growth[3]. Carbohydrate metabolism is very sensitive to changes in plant status, with alterations in fructose, glucose and sucrose concentrations occurring when stress intensity is more pronounced[6,7].
In plants, sugar production through photosynthesis is a vital process and sugar status modulates and coordinates internal regulators and environmental signals that govern growth and development. Thus, many plants use sucrose as the major form of transported carbon and it also has an important storage role. Furthermore, it is thought that sucrose and its derivates could act as regulators of cellular metabolism. All these properties are due to its non-reducing nature[8-10].
In many plant systems, one of the common features of sucrose metabolism is the continual cycling of its degradation and synthesis[11,12].The main pathway of sucrose synthesis is from hexose phosphates, which is chiefly catalysed by the combined action of the enzymes: UDP-glucosepyrophosphorylase, sucrose phosphate synthase (SPS, EC 2.4.1.14) and sucrose phosphatase (SP, EC 3.1.3.24). The reaction catalysed by SPS is readily reversible, but, as the SP step is essentially irreversible, the equilibrium for the SPS reaction is displaced and the formation of sucrose is favoured[13]. In fact, evidence suggests that SPS and SP may actually form a complex in vivo[14]. On the other hand, it has been shown that SPS is subjected to complex regulation[15]. An alternative pathway for sucrose synthesis could be from UDP-glucose and fructose, catalysed by sucrose synthase (SS, EC 2.4.1.13). In vitro, this enzyme can conduct both sucrose synthesis and degradation[13]. In sucrose-synthesising tissues, SPS activity is greater than SS activity and corresponds better to the formation rate of sucrose.
Besides sucrose synthase, plants contain other enzymes capable of cleaving sucrose: invertases (EC 3.2.1.26). Plant invertases are β-fructosidases, having acid and alkaline (neutral) forms and catalyse the irreversible hydrolysis of sucrose into glucose and fructose. It has been proposed that SS, because of its reversible character, can balance the actions of irreversible sucrose synthesis by SPS and sucrose breakdown by invertase[13]. Irreversible enzymes (SPS and invertase) generally are controlled by strong regulation, in this way, sucrose synthase allows the rate of sucrose degradation to respond sensitively and automatically to the supply of sucrose and the demand for carbon in biosynthetic and respiratory pathways in the growing cell[11,16]. Although a given tissue will tend to have an excess of one activity over the other, depending upon whether it is engaged in net sucrose synthesis or breakdown, many tissues have both enzymes and it is clear that significant “sugar cycling” can occur[17].
Calcium has been shown to ameliorate the adverse effects of salinity on plants and it is well-known that it has regulatory roles in plant metabolism[18-20]. It has been demonstrated that maize leaf SPS kinase is strictly Ca2+ dependent, raising the intriguing possibility that the cytosolic Ca2+ concentration may regulate sucrose biosynthesis, at least in some species[14]. There is evidence that cytosolic Ca2+ is decreased in the light, relative to the dark and this could contribute to the light activation of SPS in vivo[15].
The aim of the work was to determine if the influence of calcium on salinity-treated pepper plants is related to carbohydrate metabolism. For this, we studied sucrose synthase, sucrose phosphate synthase and invertases activities and changes in sucrose, glucose and fructose contents, as well as physiological parameters (relative growth rate, CO2 absorption and osmotic potential).
MATERIALS AND METHODS
Plant material and growth conditions: Pepper seeds (Capsicum annuum L. Cv. California) were pre-hydrated with aerated de-ionised water for 24 h and germinated in Petri dishes, at 28°C in an incubator for 5 days. Next, embryos were transferred to a controlled-environment chamber with a 16 h light 8 h dark cycle, with air temperatures of 25 and 20°C, respectively. The relative humidity (RH) was 60% (day) and 80% (night) and photosynthetically active radiation (PAR) was 400 μmol m-2 s-1 , provided by a combination of fluorescent tubes (Philips TLD 36 W/83, Germany and Sylvania F36 W/GRO, USA) and metal halide lamps (Osram HQI.T 400 W, Germany). After 5 days, the seedlings were placed in 4 L containers and were supplied with a complete modified Hoagland nutrient solution. The solution was replaced completely every week. After 21 days, plants were treated with 10 mM CaCl2, 50 mM NaCl, or 10 mM CaCl2+50 mM NaCl. Relative growth rate (RGR), osmotic potential (Ψπ), photosynthesis rate (ACO2), sugar concentrations (glucose, fructose and sucrose) and enzyme activities (SPS, SS, acid and neutral invertases) were measured after 4 days of treatment, when plants were 30-d-old (5 fully expanded leaves and 0.2 m high). Measurements and harvesting were done at the mid-point of the light period.
Measurement of relative growth rate (RGR): Complete plant weight was determined daily and RGR (g g-1 day-1) was calculated according to the following equation:
Where:
CO2 absorption: This was measured for fully-expanded young leaves, with a portable photosynthesis and gas exchange system (LI-COR model 6200).
Leaf and root exudates: Pepper leaves and roots were transferred separately into a Eppendorf tubes perforated at the bottom and then frozen. Each tube was put inside another, non-perforated tube, then centrifuged at 500 g for 5 min at 25°C and the exudates collected in the lower Eppendorf tube. The osmotic potential and sugar concentrations were determined for the exudates. Sap analysis was done in individual leaves, but, as no differences were found among leaves of the same plant, an average value per plant is given.
Osmotic potential: The osmolarity was measured using an automatic Micro-osmometer (Roebling mod. 13/13DR-Autocal). This was calibrated using a standard KNO3 solution of 300 mOsmol kg-1 H2O. Osmotic potential was calculated by Van’t Hoff equation:
Where:
| n | = | mOsmol; | R | = | 0.083; | T | = | temperature (°K) |
Sugar concentrations: The leaf and root exudates were injected into a Dionex DX-600 HPLC chromatograph, with a CarboPac10 column, AS40 automated sampler, LC30 chromatography oven, GP50 gradient pump and an ED50 electrochemical detector. The flow rate was 1.5 ml min-1 with an eluent of NaOH/H2O, increasing, by the curve gradient method, from 18 mM NaOH to 100 mM NaOH, at 30°C. The glucose, fructose and sucrose concentrations were measured, with PeakNet 6.1 chromatography software, by comparing peak areas with those of known standards.
Enzymatic activities
Plant material extraction procedure: Pepper leaves and roots were frozen in liquid N and stored at -70°C. Approximately 3 g of stored plant material was ground with 50 mM Hepes-NaOH buffer, pH 7.0, containing 20 mM MgCl2, 10 mM isoascorbic acid, 1mM EDTA, 2% (w/v) polyvinylpyrrolidone (PVP), 1 mM dithiothreitol (DTT) and 1 mM PMSF. The crude extract was filtered through cheese cloth; afterwards the filtrate was centrifuged for 15 min at 9000 g, at 4°C. The supernatant was desalted and low-molecular-weight compounds through Sephadex G-25 micro-columns. The extract was stored and frozen in an Eppendorf tub[21].
Determination of sucrose synthesis enzyme activities (SPS, SS): For sucrose-P-synthase (SPS), the assay reaction mechanism is:
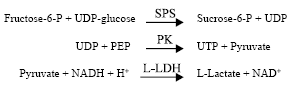 |
The sucrose-P-synthase activity was monitored by the production of UDP, then pyruvate kinase (PK) catalysed the reaction of UDP and PEP to produce UTP and pyruvate, which, in the presence of NADH, generates L-Lactate and NAD+ by the action of L-lactate dehydrogenase[21].
The disappearance of NADH by oxidation was followed spectrophotometrically. The reaction mixture contained 50 μl of extract and 950 μl of assay buffer (50 mM Hepes-NaOH pH 7.5, with 10 mM fructose-6-P, 10 mM UDP-glucose and 10 mM MgCl2). After an appropriate incubation time (20 min), the reaction was stopped by heating the test tube in boiling water for 2 min. Afterwards, when the tube contents had returned to the ambient temperature, UDP formation was assayed by adding 100 μl PEP/NADH solution (42 mg NADH-Na2, 60 mg PEP-Na and 300 mg NaHCO3 in 6 ml water) and 1.9 ml Hepes-NaOH (50 mM), pH 7.5. The initial absorbance, A0, was read at 340 nm. Then, oxidation of NADH was started by addition of 10 μl of a solution which contained 2 U of pyruvate kinase and 2 U of L-lactate dehydrogenase. Absorbance (A1) was recorded 60 min later, at 340 nm. The activity was expressed as nmol sucrose-6-P g-1f.w. min-1
Sucrose synthase activity (SS) was determined by the same protocol, but, instead of fructose-6-P, fructose was added the assay buffer. The determination of these enzyme activities was by NADH disappearance, which is proportional to the formation of UDP in sucrose synthesis. The activity was expressed as nmol sucrose g-1 f.w. min-1
Determination of sucrose hydrolysis enzyme activities (acid and neutral invertases): Invertases will, when incubated with sucrose, form glucose and fructose, thus their activities were assayed by measuring the concentration of glucose produced (expressed as nmol glucose g-1f.w. min-1)[21]. Aliquots (100 μl) of the extract were incubated for 45 min, at 30°C, with 400 μl of 50 mM Mes buffer, pH 5.5 and pH 7.5 for the acid and neutral activities, respectively, both containing 50 mM sucrose. After 45 min incubation, the reaction was stopped by adding 100 μl of 2 M NaOH to the assay mixtures (for the blanks, immediately after the start of the reaction). The concentrations of glucose and fructose were measured with anthrone[22].
RESULTS
Effects of NaCl and CaCl2 on osmotic potential, growth and CO2 assimilation: In leaves, significant differences between treatments did not appear for absolute osmotic potential values (Fig. 1). In roots, a significant increase was observed in plants treated with NaCl or NaCl + CaCl2, but the increase was higher in plants treated only with NaCl.
In Fig. 2 a significant decrease in RGR can be observed for plants treated with NaCl, compared with the control. However, the other treatments did not show significant differences.
The CO2 assimilation is represented in Fig. 3. Again, a considerable decrease in the 50 mM NaCl treatment was observed while the rest of the treatments did not exhibit significant differences with respect to the control.
Sugar content determination: Glucose, fructose and sucrose concentrations were determined in leaves and roots (Fig. 4). In leaves, monosaccharide concentrations (glucose and fructose) were increased significantly in the CaCl2 treatment, whereas for the treatments with NaCl or NaCl + CaCl2 significant changes were not revealed.
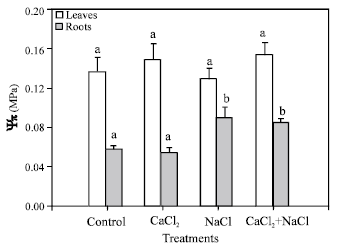 | |
| Fig. 1: | Absolute osmotic potential values of roots and leaves of pepper plants grown under different treatments (control, 10 mM CaCl2, 50 mM NaCl, 10 mM CaCl2 + 50 mM NaCl). Bars represent the means of five samples ±SE. Columns with the same letters are not significantly different (p<0.05, Tukey test). Statistical analysis has been done separately for roots and leaves |
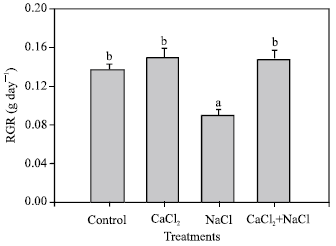 | |
| Fig. 2: | Measurement of the relative growth rate of pepper plants grown under different treatments (control, 10 mM CaCl2, 50 mM NaCl, 10 mM CaCl2 + 50 mM NaCl). Bars represent the means of five samples ±SE. Columns with the same letters are not significantly different (p<0.05, Tukey test) |
Sucrose concentration was greater than monosaccharide concentrations in control plants and in both NaCl treatments, nevertheless plants exposed to 10 mM CaCl2 showed the opposite result; higher monosaccharide concentrations and a lower sucrose concentration. In plants treated with NaCl, sucrose concentrations were decreased compared to the control plants, while there were no significant differences from concentrations found in NaCl + CaCl2 plants.
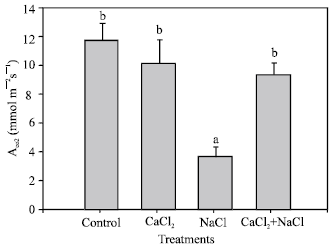 | |
| Fig. 3: | CO2 absorption of pepper plants grown under different treatments (control, 10 mM CaCl2, 50 mM NaCl, 10 mM CaCl2 + 50 mM NaCl). Bars represent the means of five samples ±SE. Columns with the same letters are not significantly different (p<0.05, Tukey test) |
In roots, only treatment with CaCl2 produced an increase of glucose and fructose concentrations and a decrease of sucrose. The rest of the treatments did not show significant differences in sugar concentrations.
Determination of sucrose synthesis enzymes (SPS and SS): The SPS activity was determined in leaves and roots (Fig. 5). Except for NaCl-treated plants, the activity of SPS was higher in leaves than in roots. Compared with the results obtained in control plants, plants treated with CaCl2 or NaCl + CaCl2 did not show any significant changes in leaves. However, in plants treated only with NaCl an important decrease in leaves was observed, although root values showed no differences.
In roots, a significant increase in SPS activity was observed in plants treated with CaCl2 and a decreased occurred in NaCl-plants. However, there were no significant differences with the NaCl + CaCl2 treatment.
The SS activity (Fig. 5) was higher than SPS activity. In both leaves and roots, there were no significant differences between the treatments, although in roots the activity was increased in plants exposed to NaCl + CaCl2.
Determination of sucrose hydrolysis enzymes (acid and neutral invertases): Sucrose hydrolysis, which occurs in the vacuole and cell wall, is catalysed by acid invertase (Fig. 6). In leaves, a decrease was found in acid invertase activity in all treatments compared with control plants. In roots, a slight increase was observed in both treatments which included calcium, compared with the control.
Neutral invertase, a cytosolic enzyme, showed an activity increment in leaves and roots of plants grown with 10 mM CaCl2 (Fig. 6).
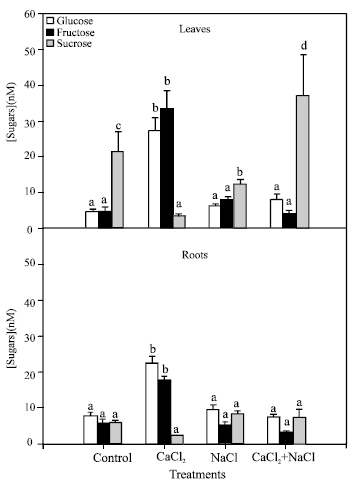 | |
| Fig. 4: | Glucose, fructose and sucrose concentrations in leaves and roots of pepper plants grown under different treatments (control, 10 mM CaCl2, 50 mM NaCl, 10 mM CaCl2 + 50 mM NaCl). Bars represent the means of twenty five samples ± SE. Columns with the same letters are not significantly different (p<0.05, Tukey test). Statistical analysis has been done separately for each sugar |
On the other hand, neutral activity at 50 mM NaCl declined significantly in roots. However, it was enhanced significantly in leaves. In the NaCl + CaCl2 treatment, a similar increase as in the CaCl2 treatment was observed, although, in this case, it was greater in roots than in leaves.
DISCUSSION
Plant osmotic adjustment involves a net accumulation of solutes in cells in response to the reduction of the medium water potential. In this way, plants maintain turgor and associated processes such as growth and development, stomatal opening and photosynthesis[1]. As a result of solutes accumulation, the osmotic potential decreases and turgor is preserved[23].
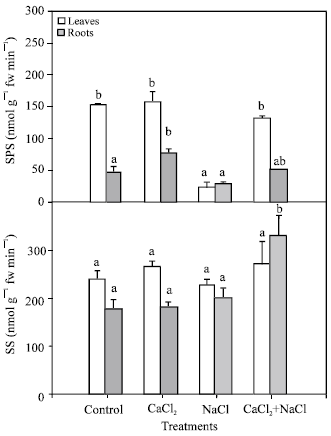 | |
| Fig. 5: | Sucrose phosphate synthase (SPS) and sucrose synthase (SS) activities in leaves and roots of pepper plants grown under different treatments (control, 10 mM CaCl2, 50 mM NaCl, 10 mM CaCl2 + 50 mM NaCl). Bars represent the means of five samples ±SE. Columns with the same letters are not significantly different (p<0.05, Tukey test). Statistical analysis has been done separately for each activity |
The results of this study indicate that, in pepper plants, osmotic potential decreased when salinity increased, but this decrease was not enough to maintain turgor and related processes, thus indicating that osmotic adjustment was not achieved. In osmotic adjustment processes, cellular osmotic potential declines due to net solutes accumulation and the turgor tends to remain steady[23,24]. Of the different studies, some relate growth with cellular turgor maintenance[25-27] some do not relate osmotic adjustment with leaf growth[28] and others show that leaf osmotic adjustment maintains growth only if plants display root osmotic adjustment[26]. On the other hand, some reports concluded that turgor is essential for development, but does not control the growth, so the water relations and osmotic adjustment determinations do not explain growth rates in plants under saline conditions[29].
In our CaCl2 treatments, a notable influence on relative growth rate (RGR) was observed.
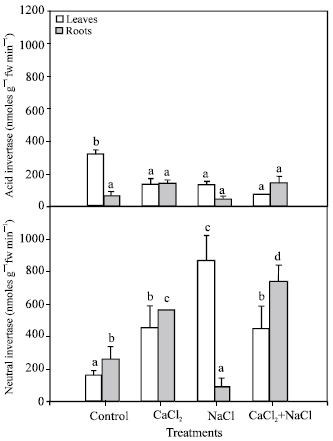 | |
| Fig. 6: | Acid and neutral invertase activities under different treatments (control, 10 mM CaCl2, 50 mM NaCl, 10 mM CaCl2 + 50 mM NaCl). Bars represent the means of five samples ±SE. Columns with the same letters are not significantly different (p<0.05, Tukey test). Statistical analysis has been done separately for each activity |
With regard to the osmotic potential, calcium, by its own, did not produce changes compared with control plants, but calcium plus 50 mM NaCl produced a rise compared with the NaCl treatment, therefore calcium could ameliorate the negative effect observed in plant water relations, as previous work has shown[19,20]. It has been described that calcium has an important role in water transport for plants exposed to salinity and can help to restore hydraulic conductivity in roots[30]. Besides, the relevance of calcium to metabolic regulation and the competition between sodium and calcium ions for binding sites of the plasma membrane are well known . Thus, it has been supposed that high levels of calcium can protect cellular membranes from the adverse effects of salinity[31,32].
When plants were exposed to moderate salinity (50 mM NaCl), the CO2 assimilation was reduced. This reduction may have been caused by damage to photosynthetic tissues, because of the accumulation of Cl-and Na+ ions[33] and by stomata closure[34]. Calcium addition to the moderately-saline medium produced a moderate increase of the CO2 absorption compared with the saline treatment. Therefore, calcium can, in some way, reduce the negative effect of NaCl on CO2 absorption. However, the mechanism is not known.
For plants grown in a saline medium, an accumulation of active osmotic solutes in tissues is needed[5]. For genotypes where salt exclusion is the major mechanism of salinity tolerance, the synthesis of organic solutes, such as sugars and amino acids, or the absorption of K+, Ca2+ or NO3-ions must increase. In energetic terms, the synthesis of organic solutes is very expensive and, thus, growth rate decreases. In other genotypes for which osmotic adjustment is the major mechanism of salinity tolerance, it is achieved by salt accumulation (mainly NaCl) in leaf tissues[35] thus increasing the cellular size, especially of the vacuole, diluting the store of salts and preventing, in this way, Na+ and Cl-accumulation in the apoplast and cytoplasm[5,36]. This could be the case for pepper plants, in previous experiments we have observed that the ion concentrations were increased, but the plants lost turgor. Thus, full osmotic adjustment, in terms of accumulation of inorganic ions and organic compounds, was not reached (Carvajal et al. unpublished results). In our salinity treatments, an increase of monosaccharide concentrations, although not very marked, was observed, despite hydrolytic activity in the cytoplasm being high. It may be that part of these monosaccharides was used not for osmotic adjustment, but for activation of alternative pathways for energy provision, such as glycolysis. It could indicate that the plant expends energy in the synthesis of monosaccharides, by activating hydrolytic enzymes, which are then used to obtain more energy, by activating glycolysis, in this way plants can grow to a higher extent. Other species more resistant to salinity demonstrate modifications of carbohydrates metabolism in saline conditions. For example, tomato plants (Lycopersicon esculentum L.) accumulated sucrose in leaves and phloem, related with an increase of SPS activity and a decrease of invertase activities[37]. On the other hand, in kikuyu grass (Pennisetum clandestinum Hochst), which is tolerant of high salinity levels, an increase in invertase activities has been observed for leaves exposed to high salinity. This points to a greater demand for request of hexoses, which control osmotic potential and cellular turgor and are needed for respiratory pathways. Glycolytic enzyme activities decrease too[3].
For roots, were not sheen significant changes during the treatments, suggesting that roots try to adjust osmotically by accumulation of other compounds. Sugar content in roots is lower than in leaves. In our experiment, when calcium was added in saline conditions, monosaccharide concentrations increased; perhaps indicating that calcium functions as a signal molecule in sucrose hydrolysis and that it could inhibit enzyme activities related to glycolysis. As in previous experiments adding Ca2+ to NaCl stressed plants, the flux of calcium into the xylem and the calcium concentration in the plasma membrane of cells was increased[19] and calcium concentration in roots and leaves sap (data not shown), we proposed a role for calcium regulating the activity of aquaporins. Also, the regulation of the enzyme SPS, by reversible phosphorylation, is mediated by SPS kinase, which is strictly Ca2+-dependent in maize roots[15]. Therefore, these observations open up the possibility that calcium could regulate sucrose biosynthesis, which is related to our results for SPS and SS activities in plants treated with NaCl+CaCl2. In any case, CaCl2 seemed to act as an antagonist of the NaCl effect on neutral invertase. However, in our plants, the results for the sucrose and hexose concentrations in CaCl2 treated plants are confusing. The increase of hexose concentration, while sucrose decreased, in CaCl2 plants suggests an effect of Ca alone that is not related to the other parameters measured.
The fact that calcium restored the inhibition of ACO2 observed in NaCl-treated plants does not explain the results obtained for sugar concentrations. In salinity treated plants, the high hydrolytic activity observed could be a consequence of the activation of alternative pathways for energy provision. In other plants (peach or chickpea), it has been reported that sugar metabolism is maintained under salinity and drought stresses[38,39]. However, in these plants, an osmotic adjustment was achieved, unlike in our pepper plants. In our leaves, NaCl could have inhibited sucrose synthesis (through its inhibition of SPS and stimulation of neutral invertase). This could explain why RGR and CO2 assimilation were affected. CaCl2 would restore RGR and CO2 by suppressing the NaCl effects on SPS and, to a lesser extent, on neutral invertase activity. Furthermore, in plants treated with CaCl2 + NaCl, the effect of calcium on SPS and SS could reveal a regulation by calcium (direct or indirect). In any case, the effect of calcium on sugar metabolism could explain its ameliorative effect of growth under saline conditions. Therefore, further research should be carried out in order to investigate the specific role of calcium in sugar metabolism of salinity stressed pepper plants.
ACKNOWLEDGMENTS
This work was supported by Instituto Euromediterraneo de Hidrotecnia (P1/01-03). The authors thank Dr. David Walker for correction of the English in the manuscript.
REFERENCES
- Greenway, H. and R. Munns, 1980. Mechanisms of salt tolerance in nonhalophytes. Annu. Rev. Plant Physiol., 31: 149-190.
CrossRefDirect Link - Ali, H., T.C. Tucher, T.L. Thompson and M. Salim, 2001. Effects of salinity and mixed ammonium and nitrate nutrition on the growth and nitrogen utilization of barley. J. Agron. Crop Sci., 186: 223-228.
Direct Link - Muscolo, A., M.R. Panuccio and M. Sidari, 2003. Effects of salinity on growth, carbohydrate metabolism and nutritive properties of kikuyu grass (Pennisetum clandestinum Hochst). Plant Sci., 164: 1103-1110.
Direct Link - Neumann, P., 1997. Salinity resistance and plant growth revisited. Plant Cell Environ., 20: 1193-1198.
CrossRefDirect Link - Oliver, M.J., P. O'Mahony and A.J. Wood, 1998. To dryness and beyond-Preparation for the dried state and rehydration in vegetative desiccation-tolerant plants. Plant Growth Regul., 24: 193-201.
CrossRefDirect Link - Balibrea, M.E., J. Dell'Amico, M.C. Bolarin and F. Perez-Alfocea, 2000. Carbon partitioning and sucrose metabolism in tomato plants growing under salinity. Physiologia Plantarum, 110: 503-511.
CrossRefDirect Link - Williams, L.E., R. Lemoine and N. Sauer, 2000. Sugar transporters in higher plants: A diversity of roles and complex regulation. Trends Plant Sci., 5: 283-290.
PubMedDirect Link - Rolland, F., B. Moore and J. Sheen, 2002. Sugar sensing and signaling in plants. Plant Cell, 2002: S185-S205.
Direct Link - Geigenberger, P. and M. Stitt, 1993. Sucrose synthase catalyses a readily reversible reaction in vivo in developing potato tubers and other plant tissues. Planta, 189: 329-339.
Direct Link - Kaya, C., H. Kirnak, D. Higgs and K. Saltali, 2002. Supplementary calcium enhances plant growth and fruit yield in strawberry cultivars grown at high (NaCl) salinity. Sci. Hortic., 93: 65-74.
CrossRef - Blum, A., R. Munns, J.B. Passioura, N.C. Turner and R.E. Sharp et al., 1996. Genetically engineered plants resistant to dry soil and salt stress: How to interpret osmotic relations. Plant Physiol., 110: 1051-1053.
Direct Link - Hasegawa, P.M., R.A. Bressan, J.K. Zhu and H.J. Bohnert, 2000. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Physiol. Plant Mol. Biol., 51: 463-499.
CrossRefDirect Link - Bolarin, M.C., M.T. Estan, M. Caro, R. Romero-Aranda and J. Cuartero, 2001. Relationship between tomato fruit growth and fruit osmotic potential under salinity. Plant Sci., 160: 1153-1159.
CrossRefDirect Link - Munns, R., 1993. Physiological processes limiting plant growth in saline soils: Some dogmas and hypotheses. Plant Cell Environ., 16: 15-24.
CrossRefDirect Link - Cramer, G.R., A. Lauchli and E. Epstein, 1986. Effects of NaCl and CaCl2 on ion activities in complex nutrient solutions and root growth of cotton. Plant Physiol., 81: 792-797.
PubMedDirect Link - Behboudian, M.H., E. Torokfalvy and R.R. Walker, 1986. Effects of salinity on ionic content, water relations and gas exchange parameters in some citrus scion-rootstock combinations. Sci. Hortic., 28: 105-116.
CrossRef - Volkmar, K.M., Y. Hu and H. Steppuhn, 1998. Physiological responses of plants to salinity: A review. Can. J. Plant Sci., 78: 19-27.
CrossRefDirect Link








