ABSTRACT
This study investigated the changes in acid invertase activity and sugar distribution during postharvest senescence in vegetable soybean (Glycine max (L.) Merr. vars. ‘Ajigen’ and ‘Huuki’) stored at 5°C for 10 days. Percent weight loss of the pods decreased with few fluctuations until the end of the experimental period. The degree of yellowing gradually increased with time. Respiration rate rapidly declined in both cultivars after 2 days of storage. The acid invertase activity of soluble and cell wall-bound fractions increased with some fluctuations throughout the storage period in both cultivars. During storage, acid invertase activity of cell wall-bound fraction showed higher activity than that of soluble fraction. There was a significant negative correlation between invertase activity (soluble and cell wall-bound fractions) and soluble sugars (sucrose, fructose and glucose). Among the three sugars, sucrose constitute the highest concentration. Glucose was present but in smaller amount than that of sucrose and fructose. Comparing the two cultivars, ‘Huuki’ showed higher percentage of weight loss, degree of yellowing, enzyme activities and sucrose content than ‘Ajigen’.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.433.438
URL: https://scialert.net/abstract/?doi=ajps.2004.433.438
INTRODUCTION
Vegetable soybean (Glycine max (L.) Merr.) also called “edamame” in Japanese (“branched bean”) and “mao dou” in Chinese (“hairy bean”), is a special vegetable originated from China more than 2000 years ago[1]. It belongs to the same specie as grain or field soybeans, but it has larger seed, sweeter flavor, smoother texture and better digestibility[2]. Worldwide, it is a minor crop but it is quite popular in East Asia. It is consumed mainly as a snack, an addition to soups or processed into sweets. Vegetable soybean has been reported to be better tasting and suitable for human consumption than grain soybean[3].
With proper storage, vegetable soybean will retain its flavor and good appearance for up to two weeks. The two most important components of flavor in vegetable soybean are sweetness and savory. Its sweet taste is determined by sucrose content and the savory taste probably by amino acid[4].
Vegetables harvested when immature, experience a disruption in energy, nutrient and hormone supplies and consequently senesce rapidly[5]. In broccoli, the first visual sign of senescence is loss of chlorophyll[6]. Sepal yellowing commences between 24 and 48 h after harvest (held at 20°C) and is essentially completed by 96 h[7].
McCollum, et al.[8] suggested that the increasing sucrose concentration in muskmelon fruit may be due to increased sucrose synthase activity accompanied by decrease in acid invertase activity during fruit growth. During handling and storage, the changes in chemical composition like sugar and organic acid contents signal the initial stage of deterioration limiting postharvest life. Normal postharvest physiology of asparagus and broccoli during air storage is characterized by the rapid loss of sucrose from the spear tips[9] and florets[10], respectively. During the first 24 h after harvest, sugars, organic acids and proteins are lost and later there are increases in the pool of free amino acids and ammonium[11,12].
Invertase plays a key role in carbohydrate metabolism and in the regulation of sucrose transport in higher plants[13]. In plants, two forms of invertase have been reported, the acid invertase, which has optimum activity at about pH 5, is located in either the vacuolar or cytosolic compartments (soluble fraction) and the insoluble extra-cellular forms which are referred to cell wall-bound fraction. The hydrolysis of sucrose by cell wall-bound invertase and subsequent import of hexoses into target cells appears to be crucial for appropriate metabolism growth and differentiation in plants[14].
There are very few reports regarding the changes in acid invertase activity and sugar distribution during postharvest senescence in vegetable soybean. Thus, this study was conducted to further understand the changes in sugar content and the influence of storage temperature at 5°C on the activity of acid invertase. Other physiological changes during storage to have a thorough knowledge of the quality of the pods after harvest, were also described.
MATERIALS AND METHODS
Plant material: Two vegetable soybean cultivars (‘Ajigen’ and ‘Huuki’) were grown in green house at Kagawa University. The pods were harvested at 42 days after anthesis and stored in perforated plastic bags with 10 holes at 5°C for 10 days. After 48 h intervals, pods were taken out from storage, shelled and stored at -30°C until analysis.
Weight loss: Weight loss was determined by weighing the seeds at random after 48 h intervals until the end of the ten-day storage period at 5°C. Weight loss was expressed as percentage of the decrease in sample weights as determined every 48 h intervals.
Color assessment: Color change in pods were determined using a chromometer (Minolta CR-200), equipped with an 8 mm measuring head. The meter was calibrated using the manufacturer’s standard white plate. Color changes were quantified in the L, a, b color spaces. L refers to the lightness of the head and ranges from black=0 to white=100. A negative value of a indicates green, while a positive number indicates red-purple color. Positive value of b indicates yellow while the negative value indicates blue color[15]. Hue angle (h° =tan-1( b/a ) when a>0 and b>0 or h°=180° + tan-1( b/a ) when a<0 and b>0 ) was calculated from the a and b values[16].
Respiration rate measurement: The pods were weighed and placed in a 6 L glass jar held at 5°C. Carbon dioxide production was measured at 48 h intervals from an intact pods sealed for 1 h by taking 10 mL gas sample from the glass jar and injected to a TCD gas chromatograph equipped with a 1 m activated charcoal column at 70°C (GC-8 AIT, Shimadzu Co. Ltd.). The result was expressed as mL CO2 kg-1 h-1.
Enzyme extraction for acid invertase: Approximately 5 g of fresh-weight seed sample were added with 1% of polyvinylpolypyrrolidone (PVPP) and 1g sea sand. The mixture was homogenized using a cooled mortar and pestle with 5 mL of 0.2 M citrate-phosphate buffer (C-P buffer) at pH 5.0. The homogenate was then filtered through four layers of cotton cloth and the filtrate was centrifuged at 12,000 x g, at 2°C for 10 min. The total supernatant was dialyzed with 0.2 M C-P buffer (pH 5.0), diluted 40 times for 12 h and the inner solution was designated as soluble fraction. The residual tissues were re-extracted with 5 mL of 0.2 M NaCl C-P buffer for about 24 h with occasional stirring. The supernatant was dialyzed as described above. The dialyzed solution was designated as cell wall-bound fraction. These extractions were carried out under 0-4°C.
Enzyme assay for acid invertase: The standard assay medium for acid invertase consisted of 0.2 M C-P buffer (pH 5.0), 0.1 mL of water and 0.1 mL of crude enzyme solution. The assay mixture was incubated at 45°C for 15 min. After the reaction, the assay mixture was neutralized with 0.1 N NaOH or 0.1 N HCl, a coloring Somogyi’s copper reagent was added and the mixture was heated for 10 min in boiling water. The amount of reducing sugars was estimated by the method of Somogyi[17]. Soluble protein content was determined by Lowry method[18] using bovine serum albumin as the standard. The enzyme activity was expressed as the amount of glucose produced per minute per milligram of protein.
Determination of sucrose, glucose and fructose contents by high performance liquid chromatography (HPLC): Approximately 2 g of fresh-weight seeds sample was mixed with 1 g of sea sand and homogenized in a cooled mortar and pestle. Ten milliliter of distilled water was added to the homogenate and centrifuged at 12,000 x g, at 2°C for 10 min. The mixture was filtered through a cellulose nitrate membrane filter (0.45 μm pore size). Soluble sugars were analyzed by HPLC using a stainless steel column (10.7 mm ID x 30 cm) packed with silica gel (gel pack C610). The mobile phase (filtered water) was pumped through the column at a flow rate of 1.0 mL min-1. The pressure was adjusted to 14-15 kg cm-2 and the temperature to 60°C. A refractive index monitor (Hitachi L-3300) was used to record the peak heights. Sucrose, glucose and fructose were identified by their retention times and were quantified according to standards.
Statistics: A randomized complete block design was adopted with three replications. The level of significance was calculated from the F value of ANOVA. The relationship between sugar content and invertase activity was described with linear correlation analysis.
RESULTS
Weight loss: Percent weight loss of two vegetable soybean cultivars during storage at 5°C for 10 d is shown in Fig. 1. Weight loss was high after 2 and 4 d of storage in ‘Huuki’ and ‘Ajigen’, respectively. A further gradual decrease in weight was observed as storage progressed in both cultivars. ‘Ajigen’ and ‘Huuki’ has a cumulative weight loss of about 10% and 19.6%, respectively, after 10 d of storage. Comparing the two cultivars, ‘Huuki’ showed higher weight loss than ‘Ajigen’.
Color/ hue angle change: Figure 2 shows the change in color of the two vegetable soybean cultivars. A continuous decrease in hue angle value was observed in both cultivars throughout the 10 d storage period. ‘Ajigen’ showed higher hue angle value than ‘Huuki’ which reflected its higher degree of greenness in the pods.
Respiration rate: A rapid decline in CO2 production was observed in both cultivars after 2 d of storage. After that it was almost unchanged until the end of the storage period (Fig. 3).
Acid invertase activity in soluble fraction: Figure 4 shows the acid invertase activity in the soluble fraction. In both cultivars, the activity increased throughout the storage period with few fluctuations. ‘Huuki’ showed higher enzyme activity than ‘Ajigen’.
Acid invertase activity in cell wall-bound fraction: Figure 5 shows the acid invertase activity in the cell wall-bound fraction. In both cultivars, invertase activity increased with some fluctuations at the end of the storage period. ‘Huuki’ showed higher activity than ‘Ajigen’.
Changes in soluble sugar content: The changes in soluble sugar content of the two vegetable soybean cultivars during storage is shown in Fig. 6. Sucrose content in ‘Ajigen’ rapidly declined after 2 d of storage and increased gradually thereafter while a gradual decreased was observed in ‘Huuki’ until the end of the 10 d storage period. There was no significant difference in fructose and glucose content in both cultivars. However, both sugars (fructose and glucose) decreased with few fluctuations until the end of the storage period. Among the three sugars, sucrose content was found significantly higher than that of fructose and glucose. Glucose was present but in smaller amount than sucrose and fructose.
Correlation coefficients (r) between acid invertase activity and sugar contents: Table 1 shows the correlation coefficient (r) between the acid invertase activity and sugar content of two vegetable soybean cultivars.
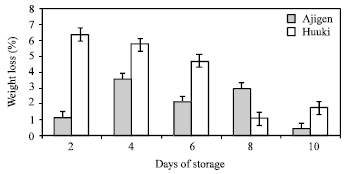 | |
| Fig. 1: | Weight loss of two vegetable soybean cultivars (‘Ajigen’ and ‘Huuki’) during storage at 5°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. |
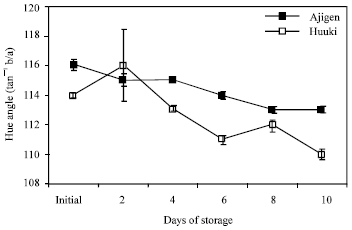 | |
| Fig. 2: | Color change of two vegetable soybean cultivars (‘Ajigen’ and ‘Huuki’) during storage at 5°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. |
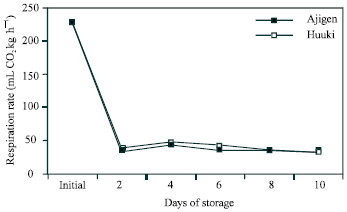 | |
| Fig. 3: | Respiration rate of two vegetable soybean cultivars (‘Ajigen’ and ‘Huuki’) during storage at 5°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. |
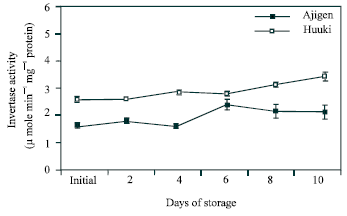 | |
| Fig. 4: | Changes in acid invertase activity in the soluble fraction of two vegetable soybean cultivars (‘Ajigen’ and ‘Huuki’) during storage at 5°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. |
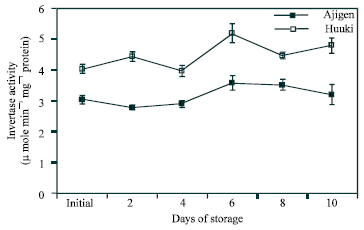 | |
| Fig. 5: | Changes in acid invertase activity in the cell wall-bound fraction of two vegetable soybean cultivars (‘Ajigen’ and ‘Huuki’) during storage at 5°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. |
| Table 1: | Correlation coefficients (r) between acid invertase activity and sugar concentration in soluble and cell wall-bound fractions of two vegetable soybean cultivars |
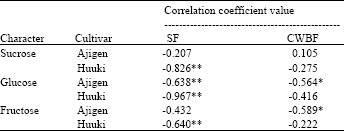 | |
| SF=soluble fraction, CWBF= cell wall-bound fraction, *,** denote significant correlation at p<0.05 and p<0.01 respectively, n=18 | |
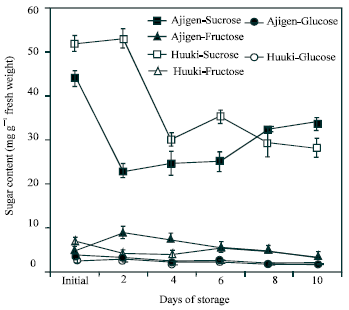 | |
| Fig. 6: | Changes in soluble sugar contents of two vegetable soybean cultivars (‘Ajigen’ and ‘Huuki’) during storage at 5°C for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate SE. SE bars were not shown when masked by the graph symbols. |
A highly significant negative correlation was observed between the acid invertase activity of soluble fraction and sugar contents (sucrose, fructose and glucose) in ‘Huuki’. There was a significant negative correlation between the acid invertase activity of cell wall-bound fraction and sugars (fructose and glucose) content in ‘Ajigen’.
DISCUSSION
There are physiological and compositional changes occurred during postharvest senescence in vegetable soybean. The principal changes that were observed include weight loss, color change, respiration rate, invertase activity and soluble sugar contents. Weight loss was high after 2 and 4 d of storage in ‘Huuki’ and ‘Ajigen’, respectively (Fig. 1). This might be a result of loss of moisture and reducing substances, which was exhibited by the shriveling of the pods. A continuous decrease in hue angle value was observed throughout the 10 d storage period which reflected decrease in greenness of the pods in both cultivars. A decline in hue angle value after harvest has previously been described in broccoli[19] could be regulated by ethylene[7,20]. A rapid decline in CO2 production was observed in both cultivars after 2 d storage period and remain unchanged at the end of the storage period (Fig. 3). This pattern of respiration rate is similar to that reported in peas and snap beans[21] and other vegetables such as asparagus[21,22]. The higher initial respiration rate in both cultivars might be due to the stress imposed by harvest.
Acid invertase activity in soluble and cell wall-bound fraction increased throughout the storage period with few fluctuations in both cultivars (Figs. 4 and 5). Similar patterns were reported by Islam et al.[23] in the CO2-enriched fruits of tomato. Coupe et al.[24] reported that soluble acid invertase activity continuously increase in broccoli florets and branchlets up to 6 d of storage. Soluble acid invertase is considered to function mainly in the breaking down of sucrose while the accumulation of sucrose into glucose and fructose. The acid invertase in the cell wall-bound fraction showed a higher activity than that of the soluble fraction throughout the experimental period. This pattern is similar to that of broccoli stored at 20°C[25] which supported our finding. Comparing the two cultivars, ‘Huuki’ has higher invertase activity (soluble and cell wall-bound fraction) than ‘Ajigen’. Sucrose was the major soluble carbohydrate in vegetable soybean during storage. Fructose and glucose were found in trace amount. Almost the three sugars started to decrease with few fluctuations from 2 d of storage and continued up to ten days (Fig. 6). The loss of sugar during storage is probably due to its transformation to cell wall material, mainly lignin and other structural substances. King and Morris[12] suggested that losses of sugars in broccoli were highest in middle and base sections during the first 12 h of storage and greatly exceeded the required amount to support CO2 production from these sections. In addition to biosynthetic processes, it is likely that some of these sugars were translocated to floral sections to help maintain the floral sugar pool and support the high respiratory demand of floral sections[19]. Conversion of starch to glucose and fructose in floral sections during the first 24 h would also contribute to maintaining the floral sugar pools.
There was a highly significant negative correlation between the acid invertase activity (soluble fraction) and sugars (sucrose, fructose and glucose) in ‘Huuki’ while a significant negative correlation was also found between the acid invertase activity (cell wall-bound fraction) and sugars (fructose and glucose) in ‘Ajigen’ (Table 1). It was assumed that the acid invertase activity had a pivotal role in postharvest metabolism by rapidly cleaving vacuolar sucrose as cytoplasmic supplies of glucose and fructose dwindled and that its rise was a response to perceived decreasing sugar levels.
Base on the results obtained, it is concluded that, there was a progressive deterioration as shown in the reduction of sugars and increase in acid invertase activity in vegetable soybean during storage at 5°C for 10 days. It also appears that percent weight loss, degree of greenness and respiration rate gradually decrease throughout the storage period. ‘Huuki’ showed higher percent of weight loss, degree of yellowing, enzyme activities and sucrose content than ‘Ajigen’. Future studies to define the role of acid invertase activity in vegetable soybean during storage associated with other sugar related enzymes are suggested.
ACKNOWLEDGMENT
The authors would like to thank the Government of Japan through its Ministry of Education, Science and Technology for the financial support.
REFERENCES
- King, G.A., D.C. Woollard, D.E. Irving and W.M. Borst, 1990. Physiological changes in asparagus spear tips after harvest. Physiol. Plant., 80: 393-400.
CrossRefDirect Link - Tian, M., C.G. Downs, R.E. Lill and G.A. King, 1994. A role for ethylene in the yellowing of broccoli after harvest. J. Am. Soc. Hortic. Sci., 119: 276-281.
Direct Link - Irving, D.E. and P.L. Hurst, 1993. Respiration, soluble carbohydrates and enzymes of carbohydrate metabolism in tips of harvested asparagus spears. Plant Sci., 94: 89-97.
Direct Link - Downs, C.G. and S.D. Somerfield, 1997. Asparagine synthase gene expression increases sucrose declines in broccoli after harvest. N. Z. J. Crop Hortic. Sci., 25: 191-195.
Direct Link - King, G.A. and S.C. Morris, 1994. Early compositional changes during postharvest senescence of broccoli. J. Am. Soc. Hortic. Sci., 119: 1000-1005.
Direct Link - Koch, K.E., 1996. Carbohydrate-modulated gene expression in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol., 47: 509-540.
CrossRefPubMedDirect Link - McGuire, R.G., 1992. Reporting of objective color measurements. HortScience, 27: 1254-1255.
CrossRefDirect Link - Lancaster, J.E., C.E. Lister, P.F. Reay and C.M. Triggs, 1997. Influence of pigment composition on skin color in a wide range of fruit and vegetables. J. Am. Soc. Hortic. Sci., 122: 594-598.
Direct Link - Platenius, H., 1942. Effect of temperature on the respiration rate and the respiratory quotient of some vegetables. Plant Physiol., 17: 179-197.
PubMedDirect Link - King, G.A., K.G. Henderson, E.M. O'Donoghue, W. Martin and R.E. Lill, 1988. Flavour and metabolic changes in asparagus during storage. Scient. Hortic., 36: 183-190.
CrossRefDirect Link - Pramanik, B.K., T. Matsui, H. Suzuki and Y. Kosugi, 2004. Changes in acid invertase activity and sugar distribution during postharvest senescence in broccoli. Pak. J. Biol. Sci., 7: 679-684.
CrossRefDirect Link








