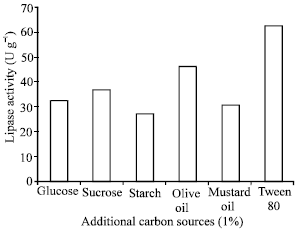
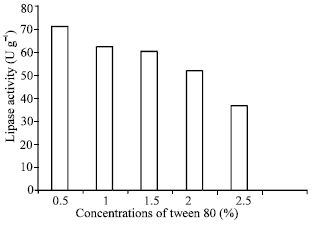
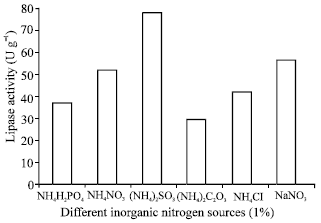
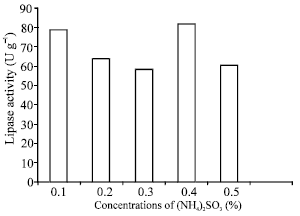
Research Article
Mineral Constituents of Culture Medium for Lipase Production by Rhizopus oligosporous Fermentation
Not Available
Kiran Shafiq
Not Available
Shazia Mirza
Not Available
Sikander Ali
Not Available
Asad-ur- Rehman
Not Available
Ikram-ul- Haq
Not Available