ABSTRACT
The effect of atrazine on the levels of carbohydrates, amino acids, potassium, phosphate and sodium contents in shoots and roots of bean plant were investigated. Application of atrazine amounting, 10 to 100 ppm resulted in decrease in carbohydrate, potassium, phosphate and sodium contents of roots and shoots of bean plants. However, amino acid content was reduced in shoot as compared to root wherein it got enhanced at 10 to 100 ppm atrazine concentration. A little growth stimulant effect was observed at 5 ppm concentration of atrazine in shoots and roots of bean plants.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2002.552.553
URL: https://scialert.net/abstract/?doi=ajps.2002.552.553
INTRODUCTION
Triazine herbicides are widely used against broad leaf weeds and crops and in tree seedling nurseries. Triazines are primarily soil applied herbicides. However, triazine can also be used for foliar with surfactants or oil. Further research showed that triazine herbicides when taken up by the root move rapidly to the top by apoplastic movement. It concentrates first in the internal veinal areas and finally in the margin of the leaf. There is no apparent synplastic movement out of the leaf (Ross and Lambi, 1985). They are photosynthetic inhibitors and cause chlorosis and desiccation of green tissues. However all these effects are observed in light and not in dark. Atrazine is a chloroamino triazine herbicide. It is a selective, pre-emergence herbicide for control of many grasses and broad leaf weeds in maize, sorghum, sugar cane and many table crops and increases the yield of crop (Shah et al., 2000). It is apoplastically translocated and leaching of atrazine is limited. Atrazine causes degradation of chloroplast and also produces swelling followed by disruption of general disk.
McClosky et al. (1990) found that atrazine concentration amounting to more than 0.05 μM inhibits protoplast O2 evolution because of binding of atrazine to photosystem II reaction. Its specific site of action is oxygen evolution or photolysis in Hill reaction of photosynthesis. It also inhibits ATP formation. Giannopolitis and Ayer (1978) reported atrazine based inhibition of photosynthesis electron transport and increase in photo-oxidation in chloroplast. Atrazine inhibits the nitrite reduction because of blockage in electron transport. Atrazine also has growth stimulant effect at low concentration in the form of increased shoot and root length, leaf blade and stem diameter. However higher concentrations result in inhibition of metabolic processes. The greening effect in atrazine treated plants is either due to increased uptake of nitrogen by the root or delay in catabolism of chlorophyll and senescence of plant. Einhelling (1989) and Raveton et al. (1997) studied the transformation and detoxification of atrazine in seedling.
The objectives of these studies were:
| a | To determine the adverse effects of atrazine on growth of bean plants, |
| b | To determine carbohydrates, amino acids, phosphate, sodium and potassium. |
MATERIALS AND METHODS
Pot experiments were conducted in growth chamber (30 °C day and 25 °C night) for one week in 2001. Five concentrations of atrazine solutions (80% WP) Viz 0, 5, 10, 50 and 100 ppm were prepared in half strength Hoagland solution (Chapman, 1976), few drops of tween-20 was used as dispersing agent. Ten to fifteen seeds of bean soaked in water for 4 hours were surface sterilized with 0.3% calcium hypochlorite for five minutes and rinsed with distilled water. They were distributed into different pots containing 0, 5, 10, 50 and 100 ppm atrazine solutions. Plants were analyzed after one week. The anthrone reactions (Tondon, 1993) are the basis of the rapid and convenient method for the determination of hexoses, aldopentoses and hexauronic acids either free or present in polysaccharides.
Carbohydrates were analyzed in protein free filtrate extract of plants in water using anthrone reagent. The complex that is formed can be estimated spectrophotometrically (Tondon, 1993). For amino acid determination, water extract of root or shoot was treated with ninhydrin solution in 10% ethanol, which was added to it and warmed at 50 to 70 °C for few minutes till purple color appeared. Optical density was measured at 566 nm (Chapman, 1976).
Sodium and potassium were estimated through dry ash method by flame photometer. For phosphate content the extracts, prepared by dry ashing method, were shaken with ammonium molybdate and stannous chloride. A blue colored complex with phosphate was obtained. Absorbance was measured at 660 nm (Chapman, 1976).
RESULTS AND DISCUSSION
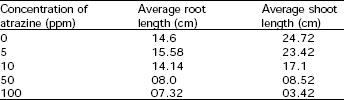
Atrazine having 10 to 100 ppm strength significantly inhibits the seedling growth. Carbohydrates, potassium, phosphate and sodium contents were decreased in shoot and root of bean plant. Amino acid got reduced in shoots, treated with atrazine at 10 to 100 ppm (Table 1), whereas in root it was increased (Table 2). The root length increased significantly at 5ppm (p< 0.05) but shoot length declined at this concentration. Both root and shoot growth was significantly retarded at concentrations of 10 ppm or more (Table 3). The slight increase in root length at 5 ppm could be due to the increased basipetal translocation of soluble carbohydrates to root while decrease in shoot length resulted because of inhibition of photosynthesis (Shaukat et al., 1975). Atrazine reduced the germination percentage (Shaukat et al., 1974 and 1975; Burhan and Shaukat, 2000). Lateral growth stimulant effect was increased with enhanced concentration of herbicide. A little growth stimulant effect was also observed at 5 ppm level.
Carbohydrates are synthesized during the process of photosynthesis and atrazine inhibits the process by degradation of chloroplast (Hill et al., 1968). Uptake of ions like sodium, potassium and phosphate presumably decreased due to the damage of vascular tissues and/or lowered energy levels in atrazine treated plants (Shaukat et al., 1975; Hence, 1985). The uptake of potassium, sodium and phosphate ions in triazine (including atrazine) treated plants has also been reported by Ebert and Dumford (1976). Potassium is involved in the protein synthesis, cell membrane and ionic balance, opening of stomata and other plant movements (Hale and Orcutt, 1987). As the herbicide influenced the uptake of potassium, which decreased as the concentration of herbicide increased (Tables 1 and 2), upward movement as well as uptake of phosphate and sodium decreased with enhanced quantity of herbicide (Tables 1 and 2).
| Table 1: | Effect of various concentrations of atrazine on carbohydrates, phosphate, sodium, potassium and amino acid content of the bean plant |
 | |
| Table 2: | Effect of various concentrations of atrazine on carbohydrates, phosphate, sodium, potassium and amino acid content of root of bean plant |
 | |
| Table 3: | Effect of atrazine on root and shoot length of bean plants |
 | |
Due to decreased rate of photosynthesis the utilization of phosphate utilization in the metabolism is also diminished and thus its rate of uptake is reduced. Atrazine inhibits the nitrite reductase activity, which is dependent on reduced ferredoxin as its electron donor. Hence blockage of electron flow to ferredoxin within the chloroplast results in accumulation of nitrite (Kelpper, 1975). Thus production of ammonia is not favored which is involved in amino acid production.
Amino acid also decreased in shoot because of the translocation of amino acid from shoot to root where as the lateral growth of root was also increased as the concentration of herbicide increased. Increased concentration of amino acid in root favors this hypothesis. Effectiveness and weed control with atrazine may be variable in the field, while some of this may be due to the climatic variation and in addition to that soil property may also be important. The herbicide is more available in gray clay than in black earth and soil pH accounted for most of the variation in the persistence of the available residues (Walker and Hargreaves, 1997).
Atrazine reduced root and shoot growth of bean plants presumably as a result of inhibition of photosynthesis. Carbohydrate and amino acid contents declined as a result of reduced synthesis while the uptake of sodium, potassium and phosphate was impeded because of reduced energy level and possibly damage of root or vascular tissues.
REFERENCES
- McClosky, W. and B. Bayer, 1990. Atrazine absorption and effect on photosynthesis in wheat leaf protoplast. Pesticide Biochem. Physiol., 37: 227-238.
Direct Link - Raveton, M., P. Ravanel, M. Kaouadji, J. Bastide and M. Tissut, 1997. The chemical transformation of atrazine in corn seedling. Pestic. Biochem. Physiol., 58: 199-208.
CrossRefDirect Link








